Advertisements
Advertisements
प्रश्न
According to VSEPR theory, the repulsion between different parts of electrons obey the order.
विकल्प
l.p – l.p > b.p–b.p> l.p–b.p
b.p–b.p> b.p–l.p> l.p–b.p
l.p–l.p> b.p–l.p > b.p–b.p
b.p–b.p> l.p–l.p> b.p–l.p
उत्तर
l.p–l.p> b.p–l.p > b.p–b.p
APPEARS IN
संबंधित प्रश्न
Stable form of A may be represented by the formula:
Explain the non-linear shape of \[\ce{H2S}\] and non-planar shape of \[\ce{PCl3}\] using valence shell electron pair repulsion theory.
Elements \[\ce{X, Y}\] and \[\ce{Z}\] have 4, 5 and 7 valence electrons respectively. Write the molecular formula of the compounds formed by these elements individually with hydrogen.
Which of the possible molecule/species is having maximum values for dipole moment. (where "A" is the central atom)?
Consider the species CH4, `"NH"_4^+` and `"BH"_4^-`. Choose the correct option with respect to these species.
The number of lone pairs of electrons on the central I atom in `"I"_3^-` is ______.
Given below are two statements: One is labelled as Assertion A and the other is labelled as Reason R:
Assertion A: The H-O-H bond angle in water molecule is 104.5°.
Reason R: The lone pair-lone pair repulsion of electrons is higher than the bond pair-bond pair repulsion.
In the light of the above statements, choose the correct answer from the options given below:
In the following structure, the percentage of the 's' character in the lone pair occupy by the oxygen atom is ______.
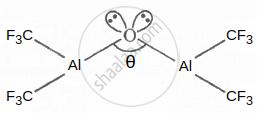
Given: Cos θ = −0.99
What is the geometry of a water molecule?
Identify compound having square pyramidal-shape:from following.
