Advertisements
Advertisements
प्रश्न
Explain the non-linear shape of \[\ce{H2S}\] and non-planar shape of \[\ce{PCl3}\] using valence shell electron pair repulsion theory.
उत्तर
The Lewis structure of \[\ce{H2S}\] is:

S-tom is surrounded by four electron pairs (two bonded and two lone pairs). These four electron pairs adopt tetrahedral arrangement. The presence of two lone pairs brings distortion in the molecule on account of repulsion with bonded pairs of electrons. Thus, the shape of \[\ce{H2S}\] molecule is V-shaped and not linear.
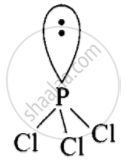
The Lewis structure of \[\ce{PCl3}\] is:
\[\begin{array}{cc}
\ce{Cl - \overset{\bullet\bullet}{P} - Cl}\\
|\phantom{.}\\
\ce{Cl}\phantom{}
\end{array}\]
P-tom is surrounded by four electron pairs (3 bonded and one lone pair). These four pairs adopt a tetrahedral geometry. Due to the presence of lone pair, \[\ce{PCl3}\] has a distorted tetrahedral geometry. Thus, it is pyramidal in shape and not non-planar shape.
APPEARS IN
संबंधित प्रश्न
Discuss the shape of the following molecules using the VSEPR model:
BeCl2, BCl3, SiCl4, AsF5, H2S, PH3
Although geometries of NH3 and H2O molecules are distorted tetrahedral, bond angle in water is less than that of ammonia. Discuss.
Explain VSEPR theory. Applying this theory to predict the shapes of IF7 and SF6.
The H-N-H bond angle in NH3 molecule is ____________.
Stable form of A may be represented by the formula:
Elements \[\ce{X, Y}\] and \[\ce{Z}\] have 4, 5 and 7 valence electrons respectively. Write the molecular formula of the compounds formed by these elements individually with hydrogen.
Elements \[\ce{X, Y}\] and \[\ce{Z}\] have 4, 5 and 7 valence electrons respectively. Which of these compounds will have the highest dipole moment?
Which of the possible molecule/species is having maximum values for dipole moment. (where "A" is the central atom)?
In the following structure, the percentage of the 's' character in the lone pair occupy by the oxygen atom is ______.
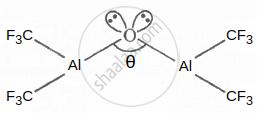
Given: Cos θ = −0.99
What is the geometry of a water molecule?
