Advertisements
Advertisements
प्रश्न
Account for the following: Fluorine does not exhibit positive oxidation state.
उत्तर
Fluorine being the most electronegative atom does not exhibit positive oxidation state because the electrons in fluorine are strongly attracted by the nuclear charge because of small size of fluorine atom and therefore, removal of an electron is not possible.
APPEARS IN
संबंधित प्रश्न
Iodine exists as
- polar molecular solid
- ionic solid
- nonpolar molecular solid
- hydrogen bonded molecular solid
Fluorine is a stronger oxidising agent than chlorine. Why?
Write two uses of ClO2.
Why are halogens coloured?
Complete chemical reactions are:Cl2 + H2O →
Complete the following equations:
KMnO4 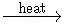
Fluorine differs from the rest of the halogens in some of its properties. This is due to ____________.
The set with the correct order of acidity is:
Which of the following statements are true?
(i) Only type of interactions between particles of noble gases are due to weak dispersion forces.
(ii) Ionisation enthalpy of molecular oxygen is very close to that of xenon.
(iii) Hydrolysis of XeF6 is a redox reaction.
(iv) Xenon fluorides are not reactive.
Give a reason for the following:
Mn+2 compounds are more stable than Fe+2 compounds.
