Advertisements
Advertisements
प्रश्न
Write two uses of ClO2.
उत्तर १
1) ClO2 is an excellent bleaching agent. It is 30 times stronger bleaching agent then the Cl2. It is used as a bleaching agerit for paper pulp in paper industry and in textile industry
2)ClO2 is also a powerful oxidising agent and chlorinating agent. It acts as a germicide for disinfecting water. It is used for purifying drinking water.
उत्तर २
Uses of ClO2:
(i) It is used for purifying water.
(ii) It is used as a bleaching agent.
APPEARS IN
संबंधित प्रश्न
Iodine exists as
- polar molecular solid
- ionic solid
- nonpolar molecular solid
- hydrogen bonded molecular solid
Draw structure of Chlorine pentafluoride
Account for the following: Fluorine does not exhibit positive oxidation state.
Compare the oxidizing action of F2 and Cl2 by considering parameters such as bond dissociation enthalpy, electron gain enthalpy and hydration enthalpy.
F2 has lower bond dissociation enthalpy than Cl2. Why?
Which halogen compound in each of the following pairs will react faster in SN2 reaction CH3Br or CH3I
Which halogen compound in each of the following pairs will react faster in SN2 reaction
(CH3)3 C – Cl or CH3 – Cl
Why are halogens coloured?
Write the reactions of F2 and Cl2 with water.
Complete chemical reactions are:Cl2 + H2O →
Complete the following equations:
KMnO4 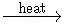
Select the correct alternative from the choices given :
The correct order of increasing acidic strength of the oxoacids of chlorine is:
The set with the correct order of acidity is:
Which is the strongest acid in the following:
In solid state \[\ce{PCl5}\] is a ______.
Reduction potentials of some ions are given below. Arrange them in decreasing order of oxidising power.
| Ion | \[\ce{CIO^{-}_{4}}\] | \[\ce{IO^{-}_{4}}\] | \[\ce{BrO^{-}_{4}}\] |
| Reduction potential EΘ/V |
EΘ = 1.19 V | EΘ = 1.65V | EΘ = 1.74 V |
Iodine has lowest oxidiation state in ______.
Statement I: Acid strength increases in the order given as HF << HCl << HBr << HI.
Statement II: As the size of the elements F, Cl, Br, and I increase down the group, the bond strength of HF, HCl, HBr and HI decreases and so the acid strength increases.
In light of the above statements, choose the correct answer from the options given below.
Give a reason for the following:
Mn+2 compounds are more stable than Fe+2 compounds.
