Advertisements
Advertisements
प्रश्न
Account for the following:
Arrange the following compounds in the increasing order of their basic strength in aqueous solution: CH3NH2,(CH3)3N,(CH3)2NH
उत्तर
The increasing order of basic strength in aqueous solution of the following amines is (CH3)3N < CH3NH2 < (CH3)2NH (Due to steric hindrance and +I effect of alky groups).
संबंधित प्रश्न
Write a short note on the following:
Carbylamine reaction
The correct increasing order of basic strength for the following compounds is ______.
(I)
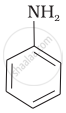
(II)
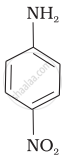
(III)
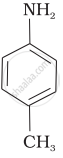
The most reactive amine towards dilute hydrochloric acid is:
The correct decreasing order of basic strength of the following species is ______.
\[\ce{H2O, NH3, OH-, NH^{-}2}\]
Explain why \[\ce{MeNH2}\] is stronger base than \[\ce{MeOH}\]?
When ethanol is mixed with ammonia and passed over alumina the compound formed is which compound?
Which of the following statement is true about methyl amine?
Which of the following is most basic?
State one reason for the following:
Alkylamine is soluble in water, whereas arylamine is insoluble in water.
The correct order of the increasing basic nature of Ammonia, Methylamine and Aniline is:
