Advertisements
Advertisements
प्रश्न
Explain why \[\ce{MeNH2}\] is stronger base than \[\ce{MeOH}\]?
उत्तर
As the electronegative of oxygen is more than the electronegativity of a nitrogen atom, the \[\ce{O - H}\] bond is more polar than the \[\ce{N - H}\] bond, therefore \[\ce{MeOH}\] is stronger acod than \[\ce{MeNH2}\] or \[\ce{MeNH2}\] 2 is stronger base than \[\ce{MeOH}\].
APPEARS IN
संबंधित प्रश्न
Arrange the following in increasing order of their basic strength in aqueous solution:
\[\ce{CH3NH2, (CH3)3N, (CH3)2NH}\]
Write the structures of main products when aniline reacts with the following reagents : HCl
Why does NH3 act as a Lewis base?
Write the structures of the main products of the following reactions:
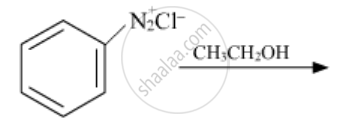
When ethanol is mixed with ammonia and passed over alumina the compound formed is which compound?
Which of the following is most basic?
By the presence of a halogen atom in the ring, what is the effect of this on basic property of aniline?
What is the characteristic smell of liquid amines?
Account for the following:
Arrange the following compounds in the increasing order of their basic strength in aqueous solution: CH3NH2,(CH3)3N,(CH3)2NH
Which of the following compound cannot be produced if 1-propane amine is treated with NaNO2 and HCl?
