Advertisements
Advertisements
प्रश्न
Account for the following.
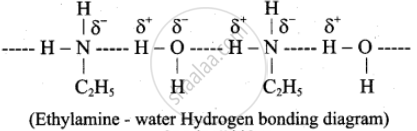
Ethylamine is soluble in water whereas aniline is not.
उत्तर
Ethylamine when added to water forms intermolecular H-bonds with water and therefore it is soluble in water. But aniline does not form an H-bond with water to a very large extent due to the presence of a large hydrophobic –C6H5 group. Hence, aniline is insoluble in water.
APPEARS IN
संबंधित प्रश्न
Choose the most correct option.
Which type of amine does produce N2 when treated with HNO2?
How are amines classified depending on the functional group? Give one example of each class of amines.
Assertion: Acetamide on reaction with KOH and bromine gives acetic acid.
Reason: Bromine catalyses hydrolysis of acetamide.
How will you convert nitrobenzene into m-nitro aniline?
Write a short note on the following.
Carbylamine reaction
Write a short note on the following.
Mustard oil reaction
Account for the following.
Gabriel phthalimide synthesis is preferred for synthesising primary amines.
How will you prepare propan-1-amine from 1-nitropropane?
(C2H5)2CHNH2. The following amine can be classified as:
Write short note on the following:
Ammonolysis
