Advertisements
Advertisements
प्रश्न
Account for the following.
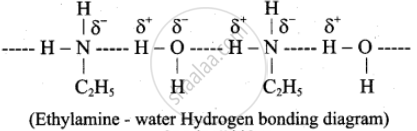
Ethylamine is soluble in water whereas aniline is not.
उत्तर
Ethylamine when added to water forms intermolecular H-bonds with water and therefore it is soluble in water. But aniline does not form an H-bond with water to a very large extent due to the presence of a large hydrophobic –C6H5 group. Hence, aniline is insoluble in water.
APPEARS IN
संबंधित प्रश्न
Classify the following amine as primary, secondary or tertiary:
(C2H5)2NH
Choose the most correct option.
Which type of amine does produce N2 when treated with HNO2?
Which of the following amines is most basic in nature in aqueous phase?
When aniline reacts with acetic anhydride the product formed is ____________.
The order of basic strength for methyl substituted amines in aqueous solution is ____________.
Write a short note on the following.
Schotten-Baumann reaction
Account for the following.
Although amino group is o- and p-directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline.
\[\begin{array}{cc}
\ce{O}\phantom{.........}\\
||\phantom{.........}\\
\ce{H - \underset{(A)}{C} - NH - CH3}
\end{array}\]
and
\[\begin{array}{cc}
\ce{O}\\
||\\
\ce{CH3 - \underset{(B)}{C} - NH2}
\end{array}\]
are which type of isomers?
Name the distinguishing test for differentiating 1° amine from 2° and 3° amine.
Write a short note on the following.
Ammonolysis
