Advertisements
Advertisements
प्रश्न
Account for the following.
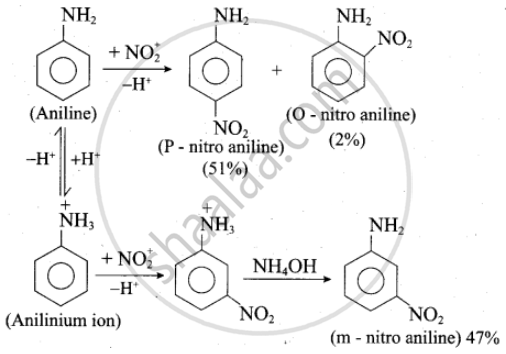
Although amino group is o- and p-directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline.
उत्तर
Nitration is usually carried out with a mixture of conc. HNO3 and conc. H2SO4. In the presence of these acids, most of the aniline gets protonated to form an anilinium ion. Therefore, in the presence of acids, the reaction mixture consists of aniline and anilinium ions.
Now –NH2, a group in aniline is O, P-directing and activating while the –NH3 group is anilinium ion is meta-directing and deactivating. Whereas nitration of aniline (due to steric hindrance at o-position) mainly gives p-nitroaniline, the nitration of anilinium ion gives m-nitro aniline. In actual practice, approximately a 1 : 1 mixture of P and m-nitro aniline is formed.
APPEARS IN
संबंधित प्रश्न
How are amines classified?
Write the number of moles of ethanoyl chloride required for complete acylation of N, N-dimethylaniline.
What is the action of nitrous acid on the following compounds?
Isopropyl amine
Account for the following.
pKb of aniline is more than that of methylamine.
Account for the following.
Gabriel phthalimide synthesis is preferred for synthesising primary amines.
How will you prepare propan-1-amine from propanamide?
Arrange the following in the increasing order of their basic strength:
C2H5NH2, C6H5NH2, (C2H5)2NH
Classify the following amine as primary, secondary or tertiary:
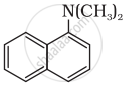
Among the following, which is the strongest base?
Account for the following:
Aniline does not undergo Friedel-Crafts reaction.
