Advertisements
Advertisements
प्रश्न
Arrange the following:
In increasing order of solubility in water:
C6H5NH2, (C2H5)2NH, C2H5NH2
उत्तर
The more extensive the H-bonding, the higher is the solubility. C2H5NH2 contains two H-atoms, whereas (C2H5)2NH contains only one H-atom. Thus, C2H5NH2 undergoes more extensive H-bonding than (C2H5)2NH. Hence, the solubility in water of C2H5NH2 is more than that of (C2H5)2NH.
Further, the solubility of amines decreases with an increase in the molecular mass. This is because the molecular mass of amines increases with an increase in the size of the hydrophobic part. The molecular mass of C6H5NH2 is greater than that of C2H5NH2 and (C2H5)2NH.
Hence, the increasing order of their solubility in water is as follows:
C6H5NH2 < (C2H5)2NH < C2H5NH2
संबंधित प्रश्न
Give reasons for the following:
CH3NH2 is more basis than C6H5NH2.
Choose the most correct option.
The hybridisation of nitrogen in primary amine is ____________.
Isobutylamine is an example of ______.
Choose the most correct option.
Identify ‘B’ in the following reactions
\[\ce{CH3 - C ≡ N ->[Na/C2H5OH] A ->[NaNO2/dilHCl]B}\]
When aniline reacts with acetic anhydride the product formed is ____________.
The order of basic strength for methyl substituted amines in aqueous solution is ____________.
Write a short note on the following:
Gabriel phthalimide synthesis
Write a short note on the following.
Carbylamine reaction
Account for the following.
pKb of aniline is more than that of methylamine.
Account for the following.
Gabriel phthalimide synthesis is preferred for synthesising primary amines.
Account for the following.
Ethylamine is soluble in water whereas aniline is not.
Arrange the following.
In decreasing order of basic strength in gas phase (C2H5)NH2, (C2H5)NH, (C2H5)3N and NH3.
Arrange the following.
In decreasing order of the pKb values C2H5NH2, C6H5NHCH3, (C2H)2NH and CH3NH2.
Arrange the following.
Increasing order of basic strength C6H5NH2, C6H5N(CH3)2, (C2H5)2NH and CH3NH2.
Arrange the following.
In decreasing order of basic strength
![]()
How will you prepare propan-1-amine from 1-nitropropane?
Identify A, B and C.
\[\ce{CH3 - NO2 ->[LiAlH4] A ->[2CH3CH2Br] B ->[H2SO4] C}\]
Classify the following amine as primary, secondary or tertiary:
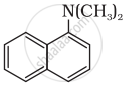
Classify the following amine as primary, secondary or tertiary:
(C2H5)2CHNH2
Write short note on the following:
Ammonolysis
Define Amines.
Write short note on the following.
Ammonolysis
Write short note on Ammonolysis.
Write short note on the following:
Ammonolysis
