Advertisements
Advertisements
Question
Account for the following.
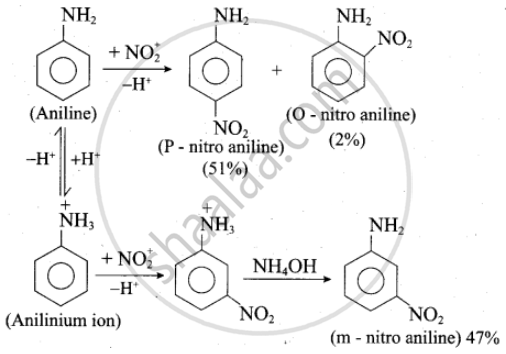
Although amino group is o- and p-directing in aromatic electrophilic substitution reactions, aniline on nitration gives a substantial amount of m-nitroaniline.
Solution
Nitration is usually carried out with a mixture of conc. HNO3 and conc. H2SO4. In the presence of these acids, most of the aniline gets protonated to form an anilinium ion. Therefore, in the presence of acids, the reaction mixture consists of aniline and anilinium ions.
Now –NH2, a group in aniline is O, P-directing and activating while the –NH3 group is anilinium ion is meta-directing and deactivating. Whereas nitration of aniline (due to steric hindrance at o-position) mainly gives p-nitroaniline, the nitration of anilinium ion gives m-nitro aniline. In actual practice, approximately a 1 : 1 mixture of P and m-nitro aniline is formed.
APPEARS IN
RELATED QUESTIONS
Choose the most correct option.
The hybridisation of nitrogen in primary amine is ____________.
Choose the most correct option.
Which type of amine does produce N2 when treated with HNO2?
The product formed by the reaction an aldehyde with a primary amine ____________.
How will you distinguish between primary secondary and tertiary aliphatic amines?
Arrange the following:
In increasing order of solubility in water:
C6H5NH2, (C2H5)2NH, C2H5NH2
Arrange the following.
Increasing order of basic strength C6H5NH2, C6H5N(CH3)2, (C2H5)2NH and CH3NH2.
Identify A, B and C.
\[\ce{CH3 - NO2 ->[LiAlH4] A ->[2CH3CH2Br] B ->[H2SO4] C}\]
Arrange the following in the increasing order of their basic strength:
C2H5NH2, C6H5NH2, (C2H5)2NH
Name the distinguishing test for differentiating 1° amine from 2° and 3° amine.
Write short note on Ammonolysis.
