Advertisements
Advertisements
प्रश्न
Amongst the following, the strongest base in aqueous medium is ______.
विकल्प
\[\ce{CH3NH2}\]
\[\ce{NCCH2NH2}\]
\[\ce{(CH3)2NH}\]
\[\ce{C6H5NHCH3}\]
उत्तर
Amongst the following, the strongest base in aqueous medium is \[\ce{(CH3)2NH}\].
Explanation:
2° amine is more basic than 1° amine, i.e., \[\ce{(CH3)2NH}\] is more basic than \[\ce{CH3NH2}\]. Due to \[\ce{^-I}\] effect of \[\ce{CN}\] group, \[\ce{NC-CH2NH2}\] is less basic than \[\ce{CH3NH2}\]. Further \[\ce{C6H5NHCH3}\] is less basic than both \[\ce{CH3NH2}\] and \[\ce{(CH3)2NH}\] due to delocalization of lone pair of electrons present on the nitrogen atom into benzene ring. Hence, the decreasing order of amines is:
\[\ce{(CH3)2NH > CH3NH2 > C6H5NHCH3 > NC – CH2NH2}\]
APPEARS IN
संबंधित प्रश्न
An aromatic compound 'A' of molecular formula C7H7ON undergoes a series of reactions as shown below. Write the structures of A, B, C, D and E in the following reactions :

Give the structures of A, B and C in the following reaction:
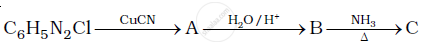
Write reactions to bring about the following conversions.
Acetamide to methylamine
Identify the product obtained when benzyl chloride undergoes ammonolysis in presence of excess ammonia followed by the reaction with two moles of methyl iodide.
For producing amines, the reaction of nitro compounds with iron scrap is preferred because:
Hoffmann Bromamide Degradation reaction is shown by ______.
In the given reaction what is the X?
\[\begin{array}{cc}
\ce{O}\phantom{.......................}\\
||\phantom{.......................}\\
\phantom{}\ce{R - C - OH <-[H3O] Χ ->[H] RCH2NH2}
\end{array}\]
When primary amines are treated with HCl, the product obtained is which of the following?
Which of the following reaction DOES NOT involve Hoffmann bromamide degradation?
Write a short note on Ammonolysis.
