Advertisements
Advertisements
प्रश्न
Amongst the following, the strongest base in aqueous medium is ______.
पर्याय
\[\ce{CH3NH2}\]
\[\ce{NCCH2NH2}\]
\[\ce{(CH3)2NH}\]
\[\ce{C6H5NHCH3}\]
उत्तर
Amongst the following, the strongest base in aqueous medium is \[\ce{(CH3)2NH}\].
Explanation:
2° amine is more basic than 1° amine, i.e., \[\ce{(CH3)2NH}\] is more basic than \[\ce{CH3NH2}\]. Due to \[\ce{^-I}\] effect of \[\ce{CN}\] group, \[\ce{NC-CH2NH2}\] is less basic than \[\ce{CH3NH2}\]. Further \[\ce{C6H5NHCH3}\] is less basic than both \[\ce{CH3NH2}\] and \[\ce{(CH3)2NH}\] due to delocalization of lone pair of electrons present on the nitrogen atom into benzene ring. Hence, the decreasing order of amines is:
\[\ce{(CH3)2NH > CH3NH2 > C6H5NHCH3 > NC – CH2NH2}\]
APPEARS IN
संबंधित प्रश्न
Write the chemical equation involved in the following reaction:
Hoffmann-bromamide degradation reaction
Which of the following amines exhibits maximum degree of intermolecular hydrogen bonding?
The reduction of alkyl cyanide with sodium and ethanol to give primary amines is, ____________.
Which of the following reagents is used in Mendius reduction reaction of alkyl cyanide?
Which of the following amines cannot be prepared by Gabriel phthalimide synthesis?
Which of the following reagents is used in Hofmann's elimination reaction of amines?
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
Which of the following reactions are correct?
(i)
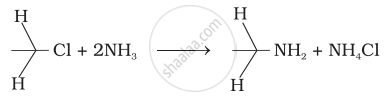
(ii)
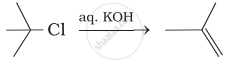
(iii)
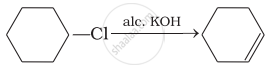
(iv)
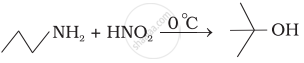
How will you carry out the following conversions?
Assertion: Only a small amount of \[\ce{HCl}\] is required in the reduction of nitro compounds with iron scrap and \[\ce{HCl}\] in the presence of steam.
Reason: \[\ce{FeCl2}\] formed gets hydrolysed to release \[\ce{HCl}\] during the reaction.
