Advertisements
Advertisements
प्रश्न
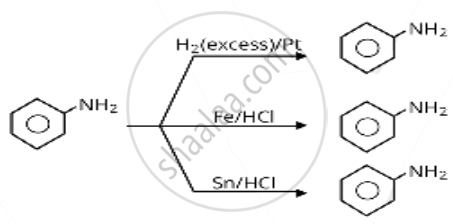
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
पर्याय
\[\ce{H2 (excess)/Pt}\]
\[\ce{LiAlH4}\] in ether
\[\ce{Fe}\] and \[\ce{HCl}\]
\[\ce{Sn}\] and \[\ce{HCl}\]
उत्तर
\[\ce{LiAlH4}\] in ether
Explanation:
Lithium aluminium hydride in either is a strong reducing agent that donated its \[\ce{H-}\] hydride ion to any \[\ce{C = O}\] containing a functional group. In addition to \[\ce{LiAlH}\], to aryl nitro compounds, no reaction will be observed, the desired products of amines will not be produced, rather it will form diazobenzene products.
\[\ce{2C4H5NO ->[LiAlH4/ether] C6H5N = N - C6H5}\]
APPEARS IN
संबंधित प्रश्न
What is the molar mass of the amine formed when acetamide undergoes Hofmann bromamide degradation?
Which of the following reagents is used in Hofmann's elimination reaction of amines?
Amongst the following, the strongest base in aqueous medium is ______.
Amongst the given set of reactants, the most appropriate for preparing 2° amine is ______.
Among the following amines, the strongest Brönsted base is:
Which of the following methods of preparation of amines will give same number of carbon atoms in the chain of amines as in the reactant?
Under which of the following reaction conditions, aniline gives p-nitro derivative as the major product?
(i) Acetyl chloride/pyridine followed by reaction with conc.\[\ce{H2SO4 }\] + conc. \[\ce{HNO3}\].
(ii) Acetic anyhdride/pyridine followed by conc.\[\ce{H2SO4}\] + conc.\[\ce{HNO3}\].
(iii) Dil. HCl followed by reaction with conc.\[\ce{H2SO4}\] + conc.\[\ce{HNO3}\].
(iv) Reaction with conc.\[\ce{HNO3}\] + conc.\[\ce{H2 SO4}\].
Assertion: Hoffmann’s bromamide reaction is given by primary amines.
Reason: Primary amines are more basic than secondary amines.
Describe Gabriel's phthalimide synthesis. (Give reaction)
Which of the following CANNOT be prepared by ammonolysis of alkyl halide?
