Advertisements
Advertisements
प्रश्न
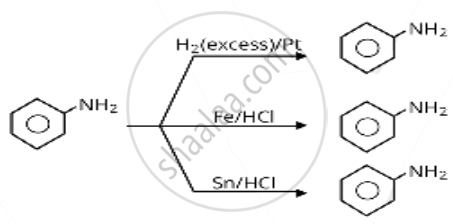
Which of the following reagents would not be a good choice for reducing an aryl nitro compound to an amine?
विकल्प
\[\ce{H2 (excess)/Pt}\]
\[\ce{LiAlH4}\] in ether
\[\ce{Fe}\] and \[\ce{HCl}\]
\[\ce{Sn}\] and \[\ce{HCl}\]
उत्तर
\[\ce{LiAlH4}\] in ether
Explanation:
Lithium aluminium hydride in either is a strong reducing agent that donated its \[\ce{H-}\] hydride ion to any \[\ce{C = O}\] containing a functional group. In addition to \[\ce{LiAlH}\], to aryl nitro compounds, no reaction will be observed, the desired products of amines will not be produced, rather it will form diazobenzene products.
\[\ce{2C4H5NO ->[LiAlH4/ether] C6H5N = N - C6H5}\]
APPEARS IN
संबंधित प्रश्न
Write a short note on the following:
Hoffmann’s bromamide reaction
Accomplish the following conversion:
Nitrobenzene to benzoic acid
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5N2Cl ->[CuCN] A ->[H2O/H+] B ->[NH3][\Delta] C}\]
Mendius reaction is used to convert _____________
Name the process of breaking C-X bond by ammonia in preparation of amines.
Which of the following reactions does NOT yield an amine?
The reagents that can be used to convert benzenediazonium chloride to benzene are:
(i) \[\ce{SnCl2/HCl}\]
(ii) \[\ce{CH3CH2OH}\]
(iii) \[\ce{H3PO2}\]
(iv) \[\ce{LiAlH4}\]
Which of the following CANNOT be prepared by ammonolysis of alkyl halide?
Which of the following amines can be prepared by Gabriel phthalimide reaction?
Write a short note on the following:
Ammonolysis
