Advertisements
Advertisements
प्रश्न
Benzylamine may be alkylated as shown in the following equation:
\[\ce{C6H5CH2NH2 + R - X -> C6H5CH2NHR}\]
Which of the following alkylhalides is best suited for this reaction through SN1 mechanism?
विकल्प
\[\ce{CH3Br}\]
\[\ce{C6H5Br}\]
\[\ce{C6H5CH2Br}\]
\[\ce{C2H5Br}\]
उत्तर
\[\ce{C6H5CH2Br}\]
Explanation:
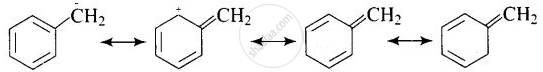
SN1 reaction occurs in two steps. In first step R – X bond is broken to produce a carbocation which is attacked by nucleophile. The greater the stability of carbocation, the greater will be the rate of reaction. Benzylic halides show high reactivity towards SN1 reaction.
APPEARS IN
संबंधित प्रश्न
Accomplish the following conversion:
Nitrobenzene to benzoic acid
Accomplish the following conversion:
Benzamide to toluene
Write the reactions of aromatic with nitrous acid.
Name the process of breaking C-X bond by ammonia in preparation of amines.
What is the molar mass of the amine formed when acetamide undergoes Hofmann bromamide degradation?
\[\ce{CH3-CN ->[Na/C2H5OH]}\]
The product formed is ____________.
The best reagent for converting 2–phenylpropanamide into 2-phenylpropanamine is ______.
Hoffmann Bromamide Degradation reaction is shown by ______.
Ethylamine can be prepared by the action of bromine and caustic potash on which compound?
Write short notes on the following:
Ammonolysis
