Advertisements
Advertisements
Question
Amongst the following, the strongest base in aqueous medium is ______.
Options
\[\ce{CH3NH2}\]
\[\ce{NCCH2NH2}\]
\[\ce{(CH3)2NH}\]
\[\ce{C6H5NHCH3}\]
Solution
Amongst the following, the strongest base in aqueous medium is \[\ce{(CH3)2NH}\].
Explanation:
2° amine is more basic than 1° amine, i.e., \[\ce{(CH3)2NH}\] is more basic than \[\ce{CH3NH2}\]. Due to \[\ce{^-I}\] effect of \[\ce{CN}\] group, \[\ce{NC-CH2NH2}\] is less basic than \[\ce{CH3NH2}\]. Further \[\ce{C6H5NHCH3}\] is less basic than both \[\ce{CH3NH2}\] and \[\ce{(CH3)2NH}\] due to delocalization of lone pair of electrons present on the nitrogen atom into benzene ring. Hence, the decreasing order of amines is:
\[\ce{(CH3)2NH > CH3NH2 > C6H5NHCH3 > NC – CH2NH2}\]
APPEARS IN
RELATED QUESTIONS
Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
Explain the mechanism of action of hydroiodic acid on 3-methylbutan-2-ol.
Give the structures of A, B and C in the following reactions :
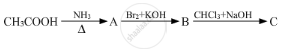
Identify compound 'B' in following series of reactions?
\[\ce{Acetonitrile ->[Na/alcohol] A ->[NaNO2/dil.HCI] B}\]
Which of the following reactions is appropriate for converting benzamide to aniline?
C6H5CONHCH3 can be converted into C6H5CH2NHCH3 by:-
- Phenyl methenamine
- N, N - Dimethylaniline
- N - Methyl aniline
- Benzenamine
Choose the correct order of the basic nature of the above amines.
Write short note on the following:
Ammonolysis
Write short note on the following.
Ammonolysis.
Assertion: Amimonolysis of alkyl halides involves the reaction between alkyl halides and alcoholic ammonia.
Reason: Ammonolysis of alkyl halides produces secondary amines only.
