Advertisements
Advertisements
प्रश्न
An organic compound having the molecular formula C3H6O can exist in the form of two isomers A and B having different functional groups. The isomer A is a liquid which is used as a solvent for nail polish. The isomer B is also a liquid. An aqueous solution of one of the lower homologues of B is used for preserving biological specimens in the laboratory
(a) What is compound A?
(b) Write the electron-dot structure of A.
(c) What is compound B?
(d) Write the electron-dot structure of B.
(e) Name the lower homologue of compound B which is used in preserving biological specimens.
उत्तर
(a) The isomer A is propanone (acetone).
(b) The electron-dot structure of propanone is::
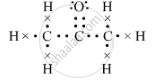
(c) The isomer B is propanal.
(d)The electron-dot structure of propanal is:
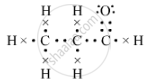
(e) Methanal or formaldehyde is the lower homologue of compound B, which is used to preserve biological specimens.
APPEARS IN
संबंधित प्रश्न
What is meant by homologous series of carbon compounds?
Classify the following carbon compounds into two homologous series and name them.
C3H4, C3H6, C4H6, C4H8, C5H8, C5H10
Write the molecular formula of first two members of homologous series having functional group −Br.
Write the molecular formula of the 2nd and 3rd member of the homologous series where the first member is ethyne.
Fill in the following blank with suitable word:
Carbon compounds have usually ... melting points and boiling points because they are ...... in nature.
Give the molecular formula of one homologue of the following:
C3H6
The molecular formula of the third member of the homologous series of ketones is:
(a) C4H8O
(b) C3H6O
(c) C5H10O
(d) C6H12O
 .
.
Find the odd one out and give its explanation.
Name the third homologue of alcohols.
