Advertisements
Advertisements
प्रश्न
Answer the following.
Explain reverse osmosis.
उत्तर
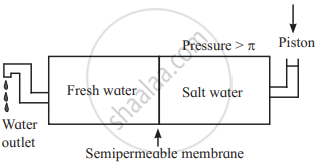
i. If a pressure larger than the osmotic pressure is applied to the solution side, then pure solvent from the solution passes into pure solvent side through the semipermeable membrane. This phenomenon is called reverse osmosis.
ii. For example, consider fresh water salt water separated by a semipermeable membrane. When the pressure larger than the osmotic pressure of a solution is applied to solution, pure water from salty water passes into fresh pure water through the membrane. Thus, the direction of osmosis can be reversed by applying a pressure larger than the osmotic pressure.
iii. The schematic set up for reverse osmosis is as follows:
APPEARS IN
संबंधित प्रश्न
Determine the osmotic pressure of a solution prepared by dissolving 2.5 × 10−2 g of K2SO4 in 2L of water at 25°C, assuming that it is completely dissociated.
(R = 0.0821 L atm K−1 mol−1, Molar mass of K2SO4 = 174 g mol−1)
Which of the following is not a colligative property?
What happens when the external pressure applied becomes more than the osmotic pressure of solution?
Blood cells are isotonic with 0.9% sodium chloride solution. What happens if we place blood cells in a solution containing
(i) 1.2% sodium chloride solution?
(ii) 0.4% sodium chloride solution?
A solution containing 15 g urea (molar mass = 60 g mol–1) per litre of solution in water has the same osmotic pressure (isotonic) as a solution of glucose (molar mass = 180 g mol–1) in water. Calculate the mass of glucose present in one litre of its solution.
Determine the osmotic pressure of a solution prepared by dissolving 25 mg of K2SO4 in 2 liter of water at 25°C, assuming that it is completely dissociated.
Define osmotic pressure.
Which of the following 0.1 M will aqueous solutions exert highest osmotic pressure?
(a) `Al_2(SO_4)_3`
(b) `Na_2SO_4`
(c) `MgCl_2`
(d) KCl
Calculate the mass of a compound (molar mass = 256 g mol−1) to be dissolved in 75 g of benzene to lower its freezing point by 0.48 K (Kf = 5.12 K kg mol−1).
Define the following term:
isotonic solution
Define the following term:
hypertonic solution
Answer the following in one or two sentences.
A solution concentration is expressed in molarity and not in molality while considering osmotic pressure. Why?
Answer the following.
What are isotonic and hypertonic solutions?
An aqueous solution of a certain organic compound has a density of 1.063 g mL-1 , osmotic pressure of 12.16 atm at 25 °C and a freezing point of 1.03 °C. What is the molar mass of the compound?
Which of the following statements is applicable for 0.1 M urea solution and 0.1 M sucrose solution?
Explain the osmotic pressure of a solution with the help of a thistle tube.
Explain the phenomenon of osmosis.
Which of the following is a colligative property?
Two solutions have different osmotic pressures. The solution of higher osmotic pressure is called ____________.
At constant temperature the osmotic pressure of a solution is ____________.
The average osmotic pressure of human blood is 7.8 bar at 37°C. What is the concentration of an aqueous NaCl solution that could be used in the blood stream?
Osmotic pressure of a solution is 0.0821 atm at a temperature of 300 K. The concentration in moles/litre will be:
The temperature at which 10% aqueous solution of (W/V) of glucose will show the osmotic pressure of 16.4 atoms is: (R = 0.082 L atom K−1 mol−1)
At a given temperature, osmotic pressure of a concentrated solution of a substance ______.
Which of the following statements is false?
Isotonic solutions must have the same:
(i) solute
(ii) density
(iii) elevation in boiling point
(iv) depression in freezing point
In isotonic solutions:
(i) Solute and solvent both are same.
(ii) Osmotic pressure is same.
(iii) Solute and solvent may or may not be same.
(iv) Solute is always same solvent may be different.
Match the items given in Column I and Column II.
| Column I | Column II |
| (i) Saturated solution | (a) Solution having same osmotic pressure at a given temperature as that of given solution. |
| (ii) Binary solution | (b) A solution whose osmotic pressure is less than that of another. |
| (iii) Isotonic solution | (c) Solution with two components. |
| (iv) Hypotonic solution | (d) A solution which contains maximum amount of solute that can be dissolved in a given amount of solvent at a given temperature. |
| (v) Solid solution | (e) A solution whose osmotic pressure is more than that of another. |
| (vi) Hypertonic solution | (f) A solution in solid phase. |
Discuss biological and industrial importance of osmosis.
How can you remove the hard calcium carbonate layer of the egg without damaging its semiprermiable membrane? Can this egg be inserted into a bottle with a narrow neck without distorting its shape? Explain the process involved.
Which one of the following is a colligative property?
Blood cells retain their normal shape in solution which are
Osmotic pressure of a solution containing 2 g dissolved protein per 300 cm3 of solution is 20 mm of Hg at 27°C. The molecular mass of protein is ______.
The following solutions were prepared by dissolving 10 g of glucose \[\ce{(C6H12O6)}\] in 250 ml of water (P1), 10 g of urea \[\ce{(CH4N2O)}\] in 250 ml of water (P2) and 10 g of sucrose \[\ce{(C12H22O11}\]) in 250 ml of water (P3). The right option for the decreasing order of osmotic pressure of these solutions is ______
Determine the osmotic pressure of a solution prepared by dissolving 2.32 × 10−2 g of K2SO4 in 2L of solution at 25°C assuming that K2SO4 is completely dissociated.
(R = 0.082 L atm K−1 mol, Molar mass K2SO4 = 174 g mol−1)
Define osmotic pressure (π).
Prove that: M2 = `(W_2RT)/(πV)`.
Define reverse osmosis.
How will you determine molar mass of solute from osmotic pressure?
Write the condition of reverse osmosis.
