Advertisements
Advertisements
प्रश्न
Arrange the following compounds in the increasing order of their densities.
(a)
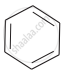
(b)
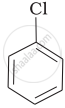
(c)
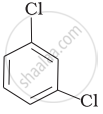
(d)
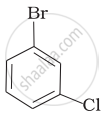
विकल्प
(a) < (b) < (c) < (d)
(a) < (c) < (d) < (b)
(d) < (c) < (b) < (a)
(b) < (d) < (c) < (a)
उत्तर
(a) < (b) < (c) < (d)
Explanation:
The density increases with increase in number of carbon atoms, halogen atoms and atomic masses of the halogen atoms bounded with the alkyl or aryl groups. Or, in other words density is related to molecular mass of the compound. Higher the molecular mass, greater will be the density of the compound.
APPEARS IN
संबंधित प्रश्न
Arrange the set of compounds in order of increasing boiling points.
Bromomethane, Bromoform, Chloromethane, Dibromomethane.
Arrange the set of compounds in order of increasing boiling points.
1-Chloropropane, Isopropyl chloride, 1-Chlorobutane.
For the same alkyl group, an alkyl bromide has a higher boiling point than alkyl fluoride because:
Which is the correct increasing order of boiling points of the following compounds?
1-Bromoethane, 1-Bromopropane, 1-Bromobutane, Bromobenzene
Out of o-and p-dibromobenzene which one has higher melting point and why?
Which of the following compounds will have the highest melting point and why?
| (I) |  |
|
(II) |
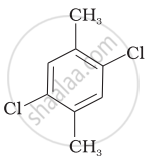 |
| (III) | 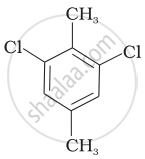 |
Arrange the following compounds in increasing order of their boiling points:
CH3CH2OH, CH3−CHO, CH3−COOH
Write the structure of the following organic halogen compound.
1,4-Dibromobut-2-ene
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
