Advertisements
Advertisements
प्रश्न
Arrange the set of compounds in order of increasing boiling points.
Bromomethane, Bromoform, Chloromethane, Dibromomethane.
उत्तर
The strength of the intermolecular interactions in organic molecules determines their boiling points. These forces consist of van der Waals forces and dipole-dipole interactions and they are dependent on the molecular mass and surface area of the molecules. Compounds with higher molecular mass have higher boiling points. Thus, the correct order is:
Chloromethane < Bromomethane < Dibromomethane < Bromoform
संबंधित प्रश्न
Explain why alkyl halides, though polar, are immiscible with water?
p-Dichlorobenzene has higher m.p. and lower solubility than those of o- and m-isomers. Discuss.
For the same alkyl group, an alkyl bromide has a higher boiling point than alkyl fluoride because:
How many structural isomers are possible for a compound with the molecular formula C3H7Cl?
Which of the following halide is 2°?
Which of the following possesses the highest melting point?
p-dichlorobenzene has a higher melting point than its o- and m-isomers because ____________.
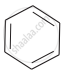
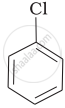
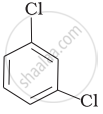
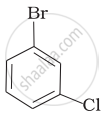
Arrange the following compounds in the increasing order of their densities.
(a)
(b)
(c)
(d)
Reaction of \[\ce{C6H5CH2Br}\] with aqueous sodium hydroxide follows ______.
Which is the correct increasing order of boiling points of the following compounds?
1-Iodobutane, 1-Bromobutane, 1-Chlorobutane, Butane
Which of the following compounds will have the highest melting point and why?
| (I) |  |
|
(II) |
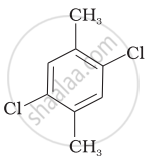 |
| (III) | 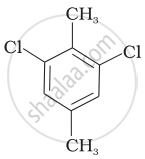 |
Which out of the following is an intensive property?
Arrange the following compounds in increasing order of their boiling points:
CH3CH2OH, CH3−CHO, CH3−COOH
Arrange the isomeric dichlorobenzene in the increasing order of their boiling point and melting points.
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
Write the structure of the following organic halogen compound.
4-tert-Butyl-3-iodoheptane
