Advertisements
Advertisements
प्रश्न
Arrange the following:
In decreasing order of basic strength in the gas phase:
C2H5NH2, (C2H5)2NH, (C2H5)3N and NH3
उत्तर
In the gas phase, there is no solvation effect. As a result, the basic strength mainly depends upon the +I effect. The higher the +I effect, the stronger is the base. Also, the greater the number of alkyl groups, the higher is the +I effect. Therefore, the given compounds can be arranged in the decreasing order of their basic strengths in the gas phase as follows:
(C2H5)3N > (C2H5)2NH > C2H5NH2 > NH3
APPEARS IN
संबंधित प्रश्न
Arrange the following:
In increasing order of basic strength:
C6H5NH2, C6H5N(CH3)2, (C2H5)2NH and CH3NH2
Write the structures of the main products of the following reactions:
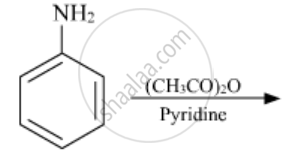
The following reaction takes place in the presence of:
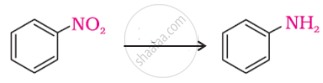
Arrange the following in decreasing order of their basic strength:
C6H5NH2, C2H5NH2, (C2H5)2NH2, NH3
The correct increasing order of basic strength for the following compounds is ______.
(I)
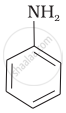
(II)
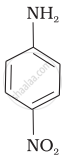
(III)
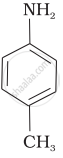
Account for the following:
Acylation of aniline is carried out in the presence of pyridine.
When ethanol is mixed with ammonia and passed over alumina the compound formed is which compound?
A Solution of methyl amine shows which type of property with litmus paper?
By the presence of a halogen atom in the ring, what is the effect of this on basic property of aniline?
Give reasons for the following observation:
pKb of aniline is lower than the m-nitroaniline.
Account for the following:
Arrange the following compounds in the increasing order of their basic strength in aqueous solution: CH3NH2,(CH3)3N,(CH3)2NH
Which of the following compound cannot be produced if 1-propane amine is treated with NaNO2 and HCl?
Arrange the decreasing order of pKb values.
\[\ce{C6H5NH2, C6H5NHCH3, C6H5CH2NH2, CH3NH2, NH3}\]
State one reason for the following:
Alkylamine is soluble in water, whereas arylamine is insoluble in water.
