Advertisements
Advertisements
प्रश्न
Arrange the following in order of increasing boiling point:
Ethoxyethane, Butanal, Butanol, n-butane
उत्तर
n-butane < ethoxyethane < butanal <butanol
APPEARS IN
संबंधित प्रश्न
Alcohols are comparatively more soluble in water than hydrocarbons of comparable molecular masses. Explain this fact.
Give simple chemical tests to distinguish between the following pairs of compounds :
Ethanol and phenol
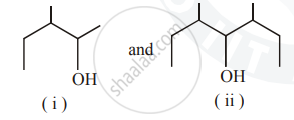
Identify the chiral molecule in the following pair :
Which statement is not correct about alcohol?
Which one of the following alcohols is least soluble in water?
Write a note on Kolbe's reaction
Convert the following:
acetaldehyde to isopropyl alcohol.
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
How is the following conversion carried out?
Methyl magnesium bromide → 2-Methylpropan-2-ol.
How is the following conversion carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
