Advertisements
Advertisements
Question
Arrange the following in order of increasing boiling point:
Ethoxyethane, Butanal, Butanol, n-butane
Solution
n-butane < ethoxyethane < butanal <butanol
APPEARS IN
RELATED QUESTIONS
Give reasons for the following: Butan-1-ol has a higher boiling point than diethyl ether.
Identify the chiral molecule in the following pair :
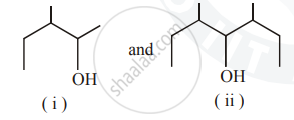
Account for the following:
CH3CHO is more reactive than CH3COCH3 towards reaction with HCN.
Write a chemical reaction to show that the open structure of D-glucose contains the following :
Five alcohol groups
Alcohols of low molecular weight are _____________.
Which statement is not correct about alcohol?
Arrange the following compounds in increasing order of boiling point. Propan-1-ol, butan-1-ol, butan-2-ol, pentan-1-ol
Arrange the following in the increasing order of their property indicated:
ethanol, ethanoic acid, benzoic acid (boiling point)
Write a note on Kolbe's reaction
How is the following conversions carried out?
\[\ce{Methyl magnesium bromide → 2-Methylpropan-2-ol}\].
