Advertisements
Advertisements
Question
Write a chemical reaction to show that the open structure of D-glucose contains the following :
Five alcohol groups
Solution
Acetylation of glucose with acetic anhydride gives glucose pentaacetate which confirms the presence of five -OH groups. Since it exists as a stable compound, five -OH groups should be attached to different carbon atoms.
\[\begin{array}{cc}
\ce{CHO} \phantom{.........................}\ce{CHO}\phantom{.........................}\\
\phantom{}|\phantom{..............................}|\phantom{............................}\\
\ce{(CHOH)4 + 5 (CH3CH)2O->(CHOCOCH3)4 + 5CH3COOH} \\
\phantom{}|\phantom{.........}\text{Acetic anhydride}\phantom{.....}|\phantom{........}\ce{O}\phantom{..................}\\
\ce{CH2OH}\phantom{........................}|\phantom{........}||\phantom{...................} \\
\text{Glucose} \phantom{........................}\ce{\underset{\text{Glucose Penta acetate}}{CH2O-C-CH3}}\phantom{.............}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Identify the chiral molecule in the following pair :
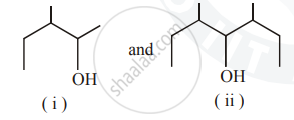
Write the structures of A, B and C in the following reactions :
\[ C_6 H_5 {NO}_2 \to^{Sn/HCI} A \to^{{NaNO}_2 /HCI}_{273 K} B \to^{H_2 O}_∆ C\]
Isopropyl alcohol is obtained by reacting which of the following alkenes with concentrated H2SO4 followed by boiling with H2O?
Assertion: Boiling points of alcohols and ethers are high.
Reason: They can form intermolecular hydrogen-bonding.
Arrange the following in the increasing order of their property indicated:
ethanol, ethanoic acid, benzoic acid (boiling point)
A solution of phenol in chloroform when treated with aqueous NaOH gives compound P as a major product. The mass percentage of carbon in P is ______. (to the nearest integer) (Atomic mass: C = 12; H = 1; O = 16)
Write a note on Kolbe's reaction
Convert the following:
acetaldehyde to isopropyl alcohol.
How is the following conversion carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
How is the following conversions carried out?
\[\ce{Methyl magnesium bromide → 2-Methylpropan-2-ol}\].
