Advertisements
Advertisements
Question
Why is the C-O bond length in phenols less than that in methanol?
Solution
The carbon-oxygen bond length in phenol (136 pm) is somewhat shorter than that in methanol. This is owing to the partial double bond character of the unshared electron pair of oxygen conjugated with the aromatic ring and the sp2 hybridised state of carbon to which oxygen is connected
APPEARS IN
RELATED QUESTIONS
\[\ce{CH3 - CH = CH2 ->[H2O/H^+]}\]
Write the structure of the product of the following reaction:
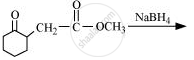
Write the structure of the compound whose IUPAC name is as follows:
2-Methylbutan-2-ol
Write the structure of the compound whose IUPAC name is as follows:
2-Ethoxy-3-methylpentane
Write the structure of the compound whose IUPAC name is as follows:
Cyclohexylmethanol
Explain why is \[\ce{O = C =O}\] nonpolar while \[\ce{R - O - R}\] is polar.
Which of the following does not form phenol or peroxide?
Cresol is:-
Ether can be used
Write the structure of the compound whose IUPAC name is as follows:
4-Chloro-3-ethylbutan-1-ol
