Advertisements
Advertisements
प्रश्न
Arrange the following in the increasing order of boiling points.
- 1-Bromopropane
- 2- Bromopropane
- 1- Bromobutane
- 1-Bromo-2-methylpropane
उत्तर
1-bromo-2-methylpropane < 2- Bromopropane
< 1-Bromopropane < 1- Bromobutane
APPEARS IN
संबंधित प्रश्न
from the following pair would undergo SN2 faster from the other?
a. ![]() b.
b. ![]()
from the following pair would undergo SN2 faster from the other?
a. CH3CH2CH2I b. CH3CH2CH2Cl
Name the reagent used to bring about the following conversion.
Bromoethane to ethoxyethane
Convert the following:
2-Chloropropane to propan-1-ol
Convert the following:
tert-Butyl bromide to isobutyl bromide
Observe the following and answer the question given below.

Can it react by SN1 mechanism? Justify your answer.
The following will react faster by SN1 mechanism
Explain. Aryl halides are less reactive than alkyl halides towards nucleophilic substitution reactions.
Explain the factors affecting SN1 and SN2 mechanism.
What is dehydrohalogenation? State the rule for the formation of the preferred product of dehydrohalogenation.
In alkaline hydrolysis of tertiary butyl bromide, ____________.
Which of the following is a primary halide?
Which of the following is least reactive towards nucleophilic addition?
Which one is most reactive towards nucleophilic addition reaction?
Which of the following is FALSE regarding SN2 reaction mechanism?
The SN2 reaction of a compound containing an asymmetric carbon atom always gives ____________.
Identify 'B' in the following reaction.
\[\ce{2-Bromobutane ->[KOH alc.][\Delta] A ->[HI] B}\]
What is the major product obtained in the sulphonation of chlorobenzene with concentrated sulphuric acid?
Explain the dehydrohalogenation reaction of 2-chlorobutane.
Observe the following and answer the question given below:
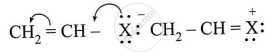
Comment on the bond length of C-X bond in it.
Discuss β - elimination reaction.
Define and explain the SN1 mechanism with a suitable example.
Complete the reaction:
\[\ce{CH3CH2Cl ->[AgCN][alc.\Delta]}\] ?
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{...............}\\
|\phantom{..................}\\
\ce{CH3 - C - CH2 - Cl ->[Na/dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{.................}
\end{array}\]
Give a reason for the following:
Ethoxy ethane with conc. HI at 373 K gives C2H5OH and CH3I but not CH3OH and C2H5I.
Complete the following reaction giving major product.
\[\begin{array}{cc}
\ce{CH3}\phantom{................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH2 - Cl->[Na/ dry ether]A}\\
|\phantom{...................}\\
\ce{CH3}\phantom{................}\\
\end{array}\]
