Advertisements
Advertisements
प्रश्न
Balance the following equation by the oxidation number method.
\[\ce{K2Cr2O7 + Kl + H2SO4 -> K2SO4 + Cr2(SO4)3 + I2 + H2O}\]
उत्तर
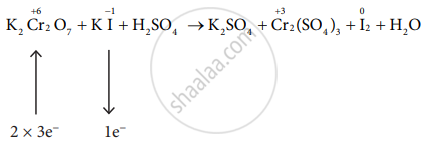
\[\ce{K2Cr2O7 + 6Kl + H2SO4 -> K2SO4 + Cr2(SO4)3 + I2 + H2O}\]
\[\ce{K2Cr2O7 + 6Kl + H2SO4 -> K2SO4 + Cr2(SO4)3 + 3I2 + H2O}\]
\[\ce{K2Cr2O7 + 6Kl + 7H2SO4 -> 4K2SO4 + Cr2(SO4)3 + 3I2 + 7H2O}\]
APPEARS IN
संबंधित प्रश्न
Choose the disproportionation reaction among the following redox reactions.
Total number of electrons present in 1.7 g of ammonia is
The correct increasing order of the oxidation state of sulphur in the anions \[\ce{SO^2-_4, SO^2-_3, S2O^2-_4, S2O^2-_6}\] is
Which of the following contains the same number of carbon atoms as in 6 g of carbon-12.
What do you understand by the term oxidation number?
Distinguish between oxidation and reduction.
Balance the following equation by the oxidation number method.
\[\ce{KMnO4 + H2C2O4 + H2SO4 -> K2SO4 + MnSO4 + CO2 + H2O}\]
Balance the following equation by the ion electron method.
\[\ce{C2O^2-_4 + Cr2O^2-_7 -> Cr^3+ + CO2}\] (in acid medium)
What is the formula of hydrolith?
In the cell represented by \[\ce{Pb(s) | Pb^{2+} (1 M) || Ag+ (1 M) | Ag(s)}\], the reducing agent is ______.
