Advertisements
Advertisements
प्रश्न
Balance the given chemical reaction as per the instructions below.
\[\ce{NaOH + H2SO4 -> Na2SO4 + H2O}\]
Write names of reactants and products of chemical reaction.
उत्तर
Names of reactants: Sodium hydroxide and Sulphuric acid
Names of products: Sodium sulphate and water
APPEARS IN
संबंधित प्रश्न
Write the balanced chemical equation for the following reaction.
\[\ce{Calcium hydroxide + Carbon dioxide → Calcium carbonate + Water}\]
2 mL of sodium hydroxide solution is added to a few pieces of granulated zinc metal taken in a test tube. When the contents are warmed, a gas evolves which is bubbled through a soap solution before testing. Write the equation of the chemical reaction involved and the test to detect the gas. Name the gas which will be evolved when the same metal reacts with dilute solution of a strong acid.
Write complete balanced equation for the following reaction:
Sulphur dioxide (gas) + Oxgyen (gas)  Sulphur trioxide (gas)
Sulphur trioxide (gas)
What is a chemical equation? Explain with the help of an example.
Carbon monoxide reacts with hydrogen under certain conditions to form methanol (CH3OH). Write a balanced chemical equation for this reaction indicating the physical states of reactants and product as well as the conditions under which this reaction takes place.
Write balanced chemical equation with state symbols for the following reaction:
Sodium hydroxide solution reacts with hydrochloric acid solution to produce sodium chloride solution and water.
What do you understand by endothermic reactions?
Is Decomposition of calcium carbonate an endothermic reaction or an exothermic reaction?
One of the following does not happen during a chemical reaction. This is:
(a) Breaking of old chemical bonds and formation of new chemical bonds
(b) Formation of new substances with entirely different properties
(c) Atoms of one element change into those of another element to form new products.
(d) A rearrangement of atoms takes place to form new products.
Balance the following chemical equation:
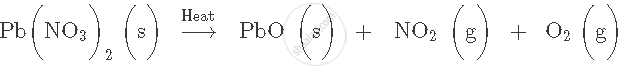
Name the type of chemical reaction shown by the following equation:
2Mg+ O2 → 2MgO
Write word equation for the following skeletal equation:
\[\ce{AgBr -> Ag + Br2}\]
What information do you get from the equation H2+ Cl2 → 2HCl ?
Balance the following equation :
Fe + O2 → Fe3 O4
Balance the following equation. Also name the product formed.
`"NaNO"_3 → "NaNO"_2 + "O"_2` Name of product (s)
Balance the following equation. Also name the product formed.
`"H"_2 +"CI"_2 → "HCI"`
Balance the following equation. Also name the product formed.
`"CO" + "O"_2 → " CO"_2`
Balance the following equation. Also name the product formed.
K2O + H2O → KOH
Write your observation and name the product when zinc reacts with dilute hydrochloric acid.
Write down what you understood from the following chemical reaction.
AgNO3(aq) + NaCI(aq) → AgCI ↓ + NaNO3(aq)
Write down what you understood from the following chemical reaction.
AgNO3(aq) + NaCI(aq) → AgCI ↓ + NaNO3(aq)
Write the answer to the following.
Explain the similarity and difference in two events, namely adding NaOH to water and adding CaO to water.
Write the chemical equation for the following word equation and balance them.
Calcium oxide + Carbon dioxide → Calcium carbonate
Balance the following equation:
Ca + N2 → Ca3N2
Complete the following blank in the equation as indicated.
\[\ce{CaH2_{(s)} + 2H2O_{( aq)}-> Ca(OH)2_{(s)} + 2H2_{(g)}}\]
Moles: 1 mole + ______ `→` ______ + ______
\[\ce{MnO2 + 4HCl -> MnCl2 + 2H2O + Cl2}\]
0.02 moles of pure MnO2 is heated strongly with conc. HCl. Calculate moles of acid required.
A chemical reaction is generally accompanied by certain external indications or characteristics. These include – change of – (a) colour (b) state (c) smell (d) evolution of gas (e) formation of precipitate (f) evolution or absorption of heat. With reference to change of colour – state the change in colour seen when the following are heated – copper carbonate.
A chemical reaction is generally accompanied by certain external indications or characteristics. These include – change of – (a) colour (b) state (c) smell (d) evolution of gas (e) formation of precipitate (f) evolution or absorption of heat. With reference to change of colour – state the change in colour seen when the following are heated – mercury [II] oxide.
In certain reaction an insoluble solid called precipitate is formed. State the colour and name of the precipitate formed in the following reaction involving addition of:
Lead nitrate to ammonium hydroxide.?
Give one example in the case where supplying energy [given below] is necessary for a chemical reaction.
Light energy
Balance the following simple equation:
ZnS + O2 → ZnO + SO2
Balance the following simple equation:
CaO + HCl → CaCl2 + H2O
Write a balanced equation for the following word equation:
Lead dioxide [lead (IV) oxide] → Lead monoxide + Oxygen
What is a chemical equation?
Three beakers labelled as A, B and C each containing 25 mL of water were taken. A small amount of NaOH, anhydrous CuSO4 and NaCl were added to the beakers A, B and C respectively. It was observed that there was an increase in the temperature of the solutions contained in beakers A and B, whereas in case of beaker C, the temperature of the solution falls. Which one of the following statement(s) is(are) correct?
- In beakers A and B, exothermic process has occurred.
- In beakers A and B, endothermic process has occurred.
- In beaker C exothermic process has occurred.
- In beaker C endothermic process has occurred.
What are the steps involved in writing the skeletal equation?
