Advertisements
Advertisements
प्रश्न
BCl3 exists as monomer whereas AlCl3 is dimerised through halogen bridging. Give reason. Explain the structure of the dimer of AlCl3 also.
उत्तर
Both the compounds, BCl3 and AlCl3 are electron-deficient compounds. In BCl3, boron is smaller in size and cannot assemble four big chlorine atoms near it causing steric hindrance and making it unstable.
Hence, BCl3 exists as a monomer only.
In AlCl3, aluminum has 3p-orbitals through which chlorine atoms can be accommodated easily to complete its octet and dimer is formed.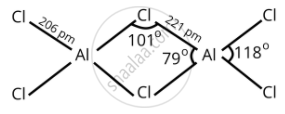
APPEARS IN
संबंधित प्रश्न
How would you explain the lower atomic radius of Ga as compared to Al?
What do you understand by inert pair effect?
The geometry of a complex species can be understood from the knowledge of type of hybridisation of orbitals of central atom. The hybridisation of orbitals of central atom in [Be(OH)4]– and the geometry of the complex are respectively.
Ionisation enthalpy (∆iH1kJ mol–1) for the elements of Group 13 follows the order.
In the structure of diborane ______.
The most commonly used reducing agent is ______.
Explain why the following compounds behave as Lewis acids?
BCl3
Aluminium dissolves in mineral acids and aqueous alkalies and thus shows amphoteric character. A piece of aluminium foil is treated with dilute hydrochloric acid or dilute sodium hydroxide solution in a test tube and on bringing a burning matchstick near the mouth of the test tube, a pop sound indicates the evolution of hydrogen gas. The same activity when performed with concentrated nitric acid, reaction doesn’t proceed. Explain the reason.
Explain the following:
Electron gain enthalpy of chlorine is more negative as compared to fluorine.
A group 13 element ‘X’ reacts with chlorine gas to produce a compound XCl3. XCl3 is electron deficient and easily reacts with NH3 to form \[\ce{Cl3X –> NH3}\] adduct; however, XCl3 does not dimerize X is ______.
