Advertisements
Advertisements
प्रश्न
BCl3 exists as monomer whereas AlCl3 is dimerised through halogen bridging. Give reason. Explain the structure of the dimer of AlCl3 also.
उत्तर
Both the compounds, BCl3 and AlCl3 are electron-deficient compounds. In BCl3, boron is smaller in size and cannot assemble four big chlorine atoms near it causing steric hindrance and making it unstable.
Hence, BCl3 exists as a monomer only.
In AlCl3, aluminum has 3p-orbitals through which chlorine atoms can be accommodated easily to complete its octet and dimer is formed.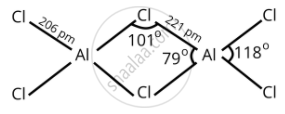
APPEARS IN
संबंधित प्रश्न
What do you understand by inert pair effect?
The geometry of a complex species can be understood from the knowledge of type of hybridisation of orbitals of central atom. The hybridisation of orbitals of central atom in [Be(OH)4]– and the geometry of the complex are respectively.
Which of the following oxides is acidic in nature?
Explain why the following compounds behave as Lewis acids?
AlCl3
When BCl3 is treated with water, it hydrolyses and forms [B[OH]4]– only whereas AlCl3 in acidified aqueous solution forms [Al(H2O)6]3+ ion. Explain what is the hybridisation of boron and aluminium in these species?
Explain the following:
PbX2 is more stable than PbX4.
Identify the compounds A, X and Z in the following reactions:
\[\ce{X ->[Δ][370 K] HBO2 ->[Δ][> 370 K] Z}\]
Describe the general trends in the following properties of the elements in Groups 13 and 14.
Atomic size
Account for the following observations:
The +1 oxidation state of thallium is more stable than its +3 state.
Which one of the following is the correct statement?
