Advertisements
Advertisements
प्रश्न
Can work be done by a system without changing its volume?
उत्तर
If the system goes through a cyclic process, then initial volume gets equal to the final volume after one cycle. But work done by the gas is non-zero. So, work can be done by a system without changing its volume.
APPEARS IN
संबंधित प्रश्न
Explain why Air pressure in a car tyre increases during driving.
When we rub our hands they become warm. Have we supplied heat to the hands?
Figure shows two processes A and B on a system. Let ∆Q1 and ∆Q2 be the heat given to the system in processes A and B respectively. Then ____________ .
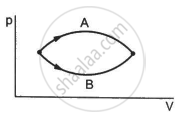
Refer to figure. Let ∆U1 and ∆U2 be the changes in internal energy of the system in the process A and B. Then _____________ .

A gas is contained in a metallic cylinder fitted with a piston. The piston is suddenly moved in to compress the gas and is maintained at this position. As time passes the pressure of the gas in the cylinder ______________ .
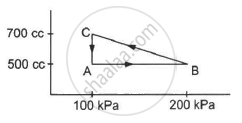
A gas is taken through a cyclic process ABCA as shown in figure. If 2.4 cal of heat is given in the process, what is the value of J ?
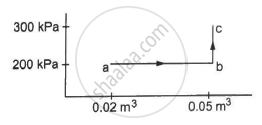
A substance is taken through the process abc as shown in figure. If the internal energy of the substance increases by 5000 J and a heat of 2625 cal is given to the system, calculate the value of J.
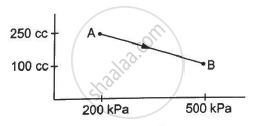
A gas is taken along the path AB as shown in figure. If 70 cal of heat is extracted from the gas in the process, calculate the change in the internal energy of the system.
A system releases 130 kJ of heat while 109 kJ of work is done on the system. Calculate the change in internal energy.
Which of the following is correct, when the energy is transferred to a system from its environment?
What is the energy associated with the random, disordered motion of the molecules of a system called as?
Explain the different ways through which the internal energy of the system can be changed.
The internal energy of a system is ______
8 m3 of a gas is heated at the pressure 105 N/m2 until its volume increases by 10%. Then, the external work done by the gas is ____________.
When 1 g of water at 0° C and 1 x 105 N/m2 pressure is converted into ice of volume 1.082 cm3, the external work done will be ____________.
Two samples A and B, of a gas at the same initial temperature and pressure are compressed from volume V to V/2; A isothermally and B adiabatically. The final pressure of A will be ______.
A gas is compressed at a constant pressure of 50 N/m2 from a volume of 10 m3 to a volume of 4 m3. Energy of 100 J is then added to the gas by heating. Its internal energy is ______.
The internal energy of one mole of argon at 300 K is ______. (R = 8.314 J/mol.K)
The molar specific heat of He at constant volume is 12.47 J/mol.K. Two moles of He are heated at constant pressure. So the rise in temperature is 10 K. Find the increase in internal energy of the gas.
What is heat?
