Advertisements
Advertisements
प्रश्न
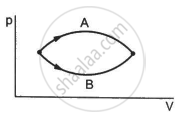
Figure shows two processes A and B on a system. Let ∆Q1 and ∆Q2 be the heat given to the system in processes A and B respectively. Then ____________ .
विकल्प
∆Q1 > ∆Q2
∆Q1 = ∆Q2
∆Q1 < ∆Q2
∆Q1 ≤ ∆Q2
उत्तर
∆Q1 > ∆Q2
Both the processes A and B have common initial and final points. So, change in internal energy, ∆U is same in both the cases. Internal energy is a state function that does not depend on the path followed.
In the P-V diagram, the area under the curve represents the work done on the system, ∆W. Since area under curve A > area under curve B, ∆W1> ∆W2.
Now,
∆Q1 = ∆U + ∆W1
∆Q2 = ∆U + ∆W2
But ∆W1 > ∆W2
⇒ ∆Q1 > ∆Q2
Here, ∆Q1 and ∆Q2 denote the heat given to the system in processes A and B, respectively.
APPEARS IN
संबंधित प्रश्न
A closed bottle contains some liquid. the bottle is shaken vigorously for 5 minutes. It is found that the temperature of the liquid is increased. Is heat transferred to the liquid? Is work done on the liquid? Neglect expansion on heating.
Can work be done by a system without changing its volume?
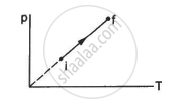
An ideal gas goes from the state i to the state f as shown in figure. The work done by the gas during the process ______________ .
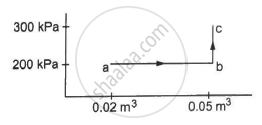
A substance is taken through the process abc as shown in figure. If the internal energy of the substance increases by 5000 J and a heat of 2625 cal is given to the system, calculate the value of J.
Which of the following is correct, when the energy is transferred to a system from its environment?
Which of the following system freely allows the exchange of energy and matter with its environment?
What is the energy associated with the random, disordered motion of the molecules of a system called as?
What is the internal energy of the system, when the amount of heat Q is added to the system and the system does not do any work during the process?
A system releases 100 kJ of heat while 80 kJ of work is done on the system. Calculate the change in internal energy.
Explain given cases related to energy transfer between the system and surrounding –
- energy transferred (Q) > 0
- energy transferred (Q) < 0
- energy transferred (Q) = 0
derive the relation between the change in internal energy (∆U), work is done (W), and heat (Q).
Which of the following represents isothermal process?
If a gas is compressed adiabatically:
The internal energy of one mole of argon at 300 K is ______. (R = 8.314 J/mol.K)
The molar specific heat of He at constant volume is 12.47 J/mol.K. Two moles of He are heated at constant pressure. So the rise in temperature is 10 K. Find the increase in internal energy of the gas.
What is heat?
Explain the change in internal energy of a thermodynamic system (the gas) by heating it.
