Advertisements
Advertisements
प्रश्न
Answer the following question:
Carbon cannot reduce the oxides of sodium, magnesium, and aluminum to their respective metals. Why? Where are these metals placed in the reactivity series? How are these metals obtained from their ores? Take an example to explain the process of extraction along with chemical equations.
उत्तर
Oxides of sodium, magnesium and aluminium are very strong oxides as these metal are very reactive metals, but carbon is not a strong reducing agent and hence cannot reduce the reactive metal oxides to metals.
In the reactivity series, sodium, magnesium and aluminium are placed in the upper portion which means these metals are very reactive in nature and carbon is less reactive.
K > Na > Ca > Mg > Al > C > Zn > Fe
Oxides of reactive metals are directly put for electrolytic reduction process to obtain the pure metal.
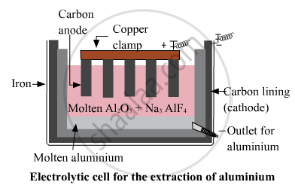
For the oxide of a reactive metal like aluminium oxide, as the metal is already in its oxide state so, it is directly put for the electrolytic reduction process. In this process, graphite electrodes are used as anode and cathode in the electrolytic chamber. The pure aluminium is attracted to the cathode, which is a lining of graphite. The oxygen is attracted to the anode, and bubbles through the solution.
Cathode reaction: at cathode reduction of aluminium takes place and thus aluminium is discharged at the cathode.
\[\ce{Al^3+ + 3e- -> Al}\]
Anode reaction: at anode oxidation takes place and oxygen gas is evolved.
\[\ce{2O^2- -> O2 + 4e-}\]
संबंधित प्रश्न
Explain giving one example, how highly reactive metals (which are high up in the reactivity series) are extracted.
Name the electrode at which aluminium metal is produced.
The metal which can be extracted from the bauxite ore is:
(a) Na
(b) Mn
(c) Al
(d) Hg
The two metals which can be extracted just by heating their sulphides in air are:
(a) sodium and copper
(b) copper and aluminium
(c) potassium and zinc
(d) mercury and copper
A sulphide ore is converted into metal oxide by the process of:
(a) carbonation
(b) roasting
(c) calcination
(d) anodising
Complete the incomplete statement with missing words:
Metals are ______ while non-metals are ______ conductors of heat.
Complete the incomplete statement with missing word:
Non-metals form acidic oxides while metals form ______.
Zinc sulphide : Roasting : : zinc carbonate : _______
Write the molecular formulae of the following compound.
Sodium aluminate
The given reaction shows one of the processes to extract the metals like Iron and Manganese.
\[\ce{MnO2(s) + Al(s) -> Mn(l) + Al2O3(s) + Heat}\]
Give a reason why Aluminium is preferably used in thermite reactions.
