Advertisements
Advertisements
प्रश्न
Choose the correct option.
The delocalization of electrons due to overlap between p orbital and sigma bond is called _______.
विकल्प
inductive effect
electronic effect
hyperconjugation
resonance
उत्तर
The delocalization of electrons due to overlap between p orbital and sigma bond is called hyperconjugation.
APPEARS IN
संबंधित प्रश्न
Identify the α-carbon in the following species and give the total number of α-hydrogens.
\[\ce{CH2 = CH - CH2 - CH3}\]
Draw a resonance structure of the following:
Acetate ion
Write true or false. Correct the false statement.
Homolytic fission involves the unsymmetrical breaking of a covalent bond.
Write true or false. Correct the false statement.
Heterolytic fission results in the formation of free radicals.
Write true or false. Correct the false statement.
Aniline is a heterocyclic compound.
Draw all the no-bond resonance structures of isopropyl carbocation.
Choose the correct option.
Which of the following statements are true with respect to electronic displacement in a covalent bond?
a. Inductive effect operates through π bond
b. Resonance effect operates through σ bond
c. Inductive effect operates through σ bond
d. Resonance effect operates through π bond
The correct IUPAC name of the compound 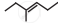 is ______.
is ______.
Choose the correct option.
The homologous series of alcohols has general molecular formula ______.
Which of the following is NOT an electrophile?
Which of the following statements is INCORRECT about hyperconjugation?
The +I inductive effect is shown by which of the following groups?
Which of the following shows positive resonance (+R) effect?
The most unstable free radical among the following is:
The overlap of σ-p orbitals is called ____________.
Which of the following is the most unstable carbocation?
How many tertiary carbon atoms and primary carbon atoms respectively are present in 2-iodo-3, 3- dimethyl pentane?
Resonance is NOT exhibited by ____________.
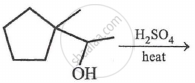
The most probable product in the reaction given below is:
Identify the group that exerts electron withdrawing resonance effect.
Identify the functional group that has an electron-donating inductive effect.
Arrange the following free radicals in order of decreasing stability.
- Methyl
- Vinyl
- Allyl
- Benzyl
Identify the α - carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
Identify the α-carbons in the following species and give the total number of α-hydrogen.
\[\ce{CH2 = CH - CH2 - CH3}\]
Identify the α - carbons in the following species and give the total number of α-hydrogen in each.
\[\ce{CH3 - CH2 - \overset{\oplus}{C}H -CH2 - CH2 }\]
Identify the α-carbons in the following species and give the total number of α-hydrogens.
\[\ce{CH3 - CH2 - \overset{⊕}{C}H - CH2 - CH3}\]
