Advertisements
Advertisements
प्रश्न
Complete the table give below.
| Element |
Atomic number |
Electron distribution | Valence electrons | Lewis dot structure |
| Lithium | 3 | 1s22s1 | ||
| Boron | 5 | 1s22s22p1 | ||
| Oxygen | 8 | 1s22s22p4 |
उत्तर
| Element |
Atomic number |
Electron distribution | Valence electrons | Lewis dot structure |
| Lithium | 3 | 1s22s1 | 1 | 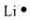 |
| Boron | 5 | 1s22s22p1 | 3 |  |
| Oxygen | 8 | 1s22s22p4 | 6 |  |
APPEARS IN
संबंधित प्रश्न
Mention the basic tendency of an atom which makes it to combine with other atoms.
Give electron dot diagram of the following:
Magnesium chloride
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2, 8, 1 |
2, 8, 7 |
2, 5 |
1 |
Answer the following question based on the table above:
What is the formula of the compound formed between W and X.
How many atoms of each kind are present in the following molecule:
Calcium oxide
State the following is a reduction reaction or oxidation reaction.
\[\ce{A^3+ + e- -> A^2+}\]
Why do they exist as monoatoms in molecules?
What do you understand by redox reactions?
State the type of bond between Y and Z and give its molecular formula.
Give the orbital diagram of the following:
Hydrogen chloride
The one which is composed of all the three kinds of bond.[ionic, covalent and coordinate bond]
