Advertisements
Advertisements
प्रश्न
Complete the table give below.
| Element |
Atomic number |
Electron distribution | Valence electrons | Lewis dot structure |
| Lithium | 3 | 1s22s1 | ||
| Boron | 5 | 1s22s22p1 | ||
| Oxygen | 8 | 1s22s22p4 |
उत्तर
| Element |
Atomic number |
Electron distribution | Valence electrons | Lewis dot structure |
| Lithium | 3 | 1s22s1 | 1 | 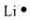 |
| Boron | 5 | 1s22s22p1 | 3 |  |
| Oxygen | 8 | 1s22s22p4 | 6 |  |
APPEARS IN
संबंधित प्रश्न
Match the atomic number 4, 8, 14, 15 and 19 with each of the following:
(a) A solid non-metal of valelncy 3.
(b) A gas of valency 2.
(c) A metal of valency 1.
(d) A non-metal of valency 4
Give electron dot diagram of the following:
nitrogen
Match the column A with Column B.
|
Column A |
Column B |
|
(i) Sodium chloride (ii) Ammonium ion (iii) Carbon tetrachloride |
Covalent bond Ionic bond Covalent and coordinate bond |
How many atoms of each kind are present in the following molecules: calcium oxide, chlorine, water, carbon tetrachloride?
How many electrons are required by each atom mentioned in (a) to attain the nearest noble gas configuration?
\[\ce{^24_12Mg}\] and \[\ce{^26_12Mg}\] are symbols of isotopes of magnesium.
(a) Compare the atoms of these isotopes with respect to :
i. the composition of their nuclei
ii. their electronic configurations
(b) Give reasons why the two isotopes of magnesium have different mass numbers.
Five atoms are labelled V to Z
| Atoms | Mass Number | Atomic Number |
| V | 40 | 20 |
| W | 19 | 9 |
| X | 7 | 3 |
| Y | 16 | 8 |
| Z | 14 | 7 |
- Which one of these atoms
(1) contains 7 protons;
(2) has an electronic configuration 2, 7? - Write down the formula of the compound formed by atoms X and Y.
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^12_6C}\]
The element that would form anion by gaining electrons in a chemical reaction is ______.
______ theory explains the formation of molecules.
Complete the following table:
| a. | Dativebond | ______ |
| b. | CaH2 → Ca+H2 | ______ |
| c. | ______ | Reduction |
| d. | ______ | Redox reaction |
