Advertisements
Advertisements
प्रश्न
\[\ce{^24_12Mg}\] and \[\ce{^26_12Mg}\] are symbols of isotopes of magnesium.
(a) Compare the atoms of these isotopes with respect to :
i. the composition of their nuclei
ii. their electronic configurations
(b) Give reasons why the two isotopes of magnesium have different mass numbers.
उत्तर
(a) i.
|
|
\[\ce{^24_12Mg}\] |
\[\ce{^26_12Mg}\] |
|
|
|
|
|
No. of electrons |
12 |
12 |
|
No. of protons |
12 |
12 |
|
No. of neutrons |
24 - 12 = 12 |
26 - 12 = 14 |
Hence, the composition of nuclei
\[\ce{_12Mg^24->(P,n),(12+12)_12Mg^26->(p,n)(12+14)}\]
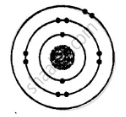
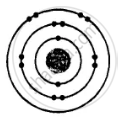
ii. Electronic configuration = 2, 8, 2
(b) Mass numbers of two isotopes of magnesium are different because of the different number of neutrons, i.e. 12 and 14, respectively.
संबंधित प्रश्न
How do atoms attain noble gas configuration?
Mention the basic tendency of an atom which makes it to combine with other atoms.
In term of electron transfer, define Oxidation
How many atoms of each kind are present in the following molecules: calcium oxide, chlorine, water, carbon tetrachloride?
How many electrons are required by each atom mentioned in (a) to attain the nearest noble gas configuration?
How many atoms of each kind are present in the following molecule:
Calcium oxide
Elements X, Y, and Z have atomic numbers 6, 9 and 12 respectively. Which one forms a cation?
Five atoms are labeled A to E
|
Atoms |
Mass No |
Atomic No. |
|
A |
40 |
20 |
|
B |
19 |
9 |
|
C |
7 |
3 |
|
D |
16 |
8 |
|
E |
14 |
7 |
(a) Which one of these atoms:
(i) contains 7 protons
(ii) has electronic configuration 2,7
(b) Write down the formula in the compound formed between C and D
(c) Predict : (i) metals (ii) non-metals
Give a reason
Physical properties of isotopes are different.
Why are Noble gases inert in nature?
Choose the odd one out and write the reason: