Advertisements
Advertisements
Question
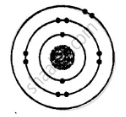
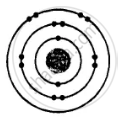
\[\ce{^24_12Mg}\] and \[\ce{^26_12Mg}\] are symbols of isotopes of magnesium.
(a) Compare the atoms of these isotopes with respect to :
i. the composition of their nuclei
ii. their electronic configurations
(b) Give reasons why the two isotopes of magnesium have different mass numbers.
Solution
(a) i.
|
|
\[\ce{^24_12Mg}\] |
\[\ce{^26_12Mg}\] |
|
|
|
|
|
No. of electrons |
12 |
12 |
|
No. of protons |
12 |
12 |
|
No. of neutrons |
24 - 12 = 12 |
26 - 12 = 14 |
Hence, the composition of nuclei
\[\ce{_12Mg^24->(P,n),(12+12)_12Mg^26->(p,n)(12+14)}\]
ii. Electronic configuration = 2, 8, 2
(b) Mass numbers of two isotopes of magnesium are different because of the different number of neutrons, i.e. 12 and 14, respectively.
RELATED QUESTIONS
In term of electron transfer, define
Reduction
What is an α (alpha) particle?
Name the following:
Two atoms having the same number of protons and electrons but different number of neutrons.
Name the following:
The shell closest to the nucleus of an atom
State the type of bonding in the following molecules.
Hydrogen chloride
Explain Octet rule with an example.
______ theory explains the formation of molecules.
Spot the error/correct the wrong statement:
In the formation of compounds, the inner shell electrons of an atom involved in bonding.
Match the following:
| 1. | Atomic bond | a. | Oxygen and hydrogen |
| 2. | Atoms with different electronegativities | b. | acceptor bond |
| 3. | An atom that accepts electron pair | c. | covalent bond |
| 4. | Rusting of iron | d. | donor atom |
| 5. | An atom that provides electron pair | e. | oxidation |
| f. | ionic bond |
How many atoms of each kind are present in the following molecule:
Chlorine