Advertisements
Advertisements
प्रश्न
\[\ce{^24_12Mg}\] and \[\ce{^26_12Mg}\] are symbols of isotopes of magnesium.
(a) Compare the atoms of these isotopes with respect to :
i. the composition of their nuclei
ii. their electronic configurations
(b) Give reasons why the two isotopes of magnesium have different mass numbers.
उत्तर
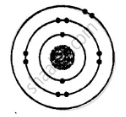
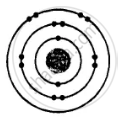
(a) i.
|
|
\[\ce{^24_12Mg}\] |
\[\ce{^26_12Mg}\] |
|
|
|
|
|
No. of electrons |
12 |
12 |
|
No. of protons |
12 |
12 |
|
No. of neutrons |
24 - 12 = 12 |
26 - 12 = 14 |
Hence, the composition of nuclei
\[\ce{_12Mg^24->(P,n),(12+12)_12Mg^26->(p,n)(12+14)}\]
ii. Electronic configuration = 2, 8, 2
(b) Mass numbers of two isotopes of magnesium are different because of the different number of neutrons, i.e. 12 and 14, respectively.
संबंधित प्रश्न
Give electron dot diagram of the following:
Magnesium chloride
Give electron dot diagram of the following:
methane
Element X is a metal with a valency 2; Y is a non-metal with a valency 3.
(a) Write an equation to show how Y forms as ion
(b) If Y is a diatomic gas, write an equation for the direct combination of X and Y to form a compound.
State the following is a reduction reaction or oxidation reaction.
\[\ce{Pb -> Pb^2+ + 2e-}\]
How do atoms attain noble gas configurations?
Draw orbit structure diagram of sodium chloride (NaCl).
Potassium (at No.19) and chlorine (at No.17) react to form a compound. Explain the formation of the compound on the basis of oxidation.
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^12_6C}\]
Explain Octet rule with an example.
How many electrons are required or released by each atom in water molecule to attain the nearest noble gas configuration?