Advertisements
Advertisements
प्रश्न
Five atoms are labelled V to Z
| Atoms | Mass Number | Atomic Number |
| V | 40 | 20 |
| W | 19 | 9 |
| X | 7 | 3 |
| Y | 16 | 8 |
| Z | 14 | 7 |
- Which one of these atoms
(1) contains 7 protons;
(2) has an electronic configuration 2, 7? - Write down the formula of the compound formed by atoms X and Y.
उत्तर
(i) As Z has atomic number of 7, hence it has 7 protons. Electronic configuration of 2, 7 means the atom has 2 + 7 = 9 electrons. Therefore, element W has electronic configuration 2, 7 since its atomic number is 9.
(ii) Number of electron in X is 3. Therefore, electronic configuration = (2, 1). As X will try to lose 1 electron to attain stable state, hence, X has valency [1+]
Number of electron in Y is 8. Therefore, electronic configuration = (2, 6). As Y will try to gain 2 electrons to attain stable state, hence, Y has valency [2−]
Since the valency of X is 1+ and valency of Y is 2−
Formula of the compound:
X1+ Y2−
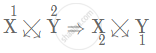
So, we get the formula as X2Y.
APPEARS IN
संबंधित प्रश्न
Mention the basic tendency of an atom which makes it to combine with other atoms.
The following table shows the electronic configuration of the elements W, X, Y, Z:
|
Element |
W |
X |
Y |
Z |
|
Electronic |
2,8,1 |
2,8,7 |
2,5 |
1 |
Answer the following questions based on the table above:
What is the formula of the compound formed between X and Z.
Explain
Octet rule for the formation of sodium chloride
An element M burns in oxygen to form an ionic compound MO. Write the formula of the compounds formed if this element is made to continue with chlorine and sulfur separately.
Identify the following reaction as either oxidation or reduction:
K - e- → K+
Identify the following reaction as either oxidation or reduction:
Zn → Zn2+ + 2e-
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^23_11Na}\]
Draw the geometric atomic structure of the following atom showing the number of electrons, protons and neutrons in them:
\[\ce{^31_15P}\]
Match the following:
| 1. | Monoatomic gaseous atom | a. | Electrovalent bond |
| 2. | Octet rule | b. | Benzene |
| 3. | Ionic bond | c. | Water |
| 4. | Non-polar solvent | d. | Electronic theory of valence |
| 5. | Polar solvent | e. | Noble gases |
Choose the odd one out and write the reason:
