Advertisements
Advertisements
प्रश्न
Consider a thin target (10–2 cm square, 10–3 m thickness) of sodium, which produces a photocurrent of 100 µA when a light of intensity 100W/m2 (λ = 660 nm) falls on it. Find the probability that a photoelectron is produced when a photons strikes a sodium atom. [Take density of Na = 0.97 kg/m3].
उत्तर
According to the problem, the area of the target A = 10–2 cm2 = 10–4 m2
And thickness, d = 10–3 m
Photocurrent, i = 100 × 10–6 A= 10–4 A
Intensity, I = 100 W/m2
⇒ λ = 660 nm = 660 × 10–9 m
ρNa = 0.97 kg/m3
Avogadro number = 6 × 1026 kg atom
Volume of sodium target = A × d
= 10–4 × 10–3
= 10–7 m3
We know that 6 × 1026 atoms of sodium weigh = 23 kg
Density of sodium = 0.97 kg/m3
Hence the volume of 6 × 1026 sodium atoms = `23/0.97` m3
Volume occupied by one sodium atom = `23/(0.97 xx (6 xx 10^36))` = 3.95 × 10–26 m3
Number of sodium atoms in target `(N_"sodium") = 10^-7/(3.95 xx 10^-26)` = 2.53 × 1018
Let m be the number of photons falling per second on the target.
Energy of each photon = `(hc)/A`
Total energy falling per second on target = `(nhc)/λ = IA`
∴ `n = (IAλ)/(hc)`
= `(100 xx 10^-4 xx (660 xx 10^-9))/((6.62 xx 10^-34) xx (3 xx 10^8))` = 3.3 × 1016
Let P be the probability of emission per atom per photon. The number of photoelectrons emitted per second
`N = P xx n xx (N_"sodium")`
= `P xx (33 xx 10^16) xx (2.53 xx 10^18)`
Now, according to the question,
i = 100 µA = 100 × 10–6 = 10–4 A
Current, i = Ne
∴ `10^-4 xx P xx (3.3 xx 10^16) xx (2.53 xx 10^18) xx (1.6 xx 10^-19)`
⇒ `P = 10^-4/((3.3 xx 10^16) xx (2.53 xx 10^18) xx (1.6 xx 10^-19))`
= 7.48 × 10–21
Then, the probability of photoemission by a single photon on a single atom is very much less than 1. Because the absorption of two photons by an atom is negligible.
APPEARS IN
संबंधित प्रश्न
Define the term 'intensity of radiation' in terms of photon picture of light.
Ultraviolet light of wavelength 2271 Å from a 100 W mercury source irradiates a photo-cell made of molybdenum metal. If the stopping potential is −1.3 V, estimate the work function of the metal. How would the photo-cell respond to a high intensity (∼105 W m−2) red light of wavelength 6328 Å produced by a He-Ne laser?
Can a photon be deflected by an electric field? Or by a magnetic field?
Should the energy of a photon be called its kinetic energy or its internal energy?
When the intensity of a light source in increased,
(a) the number of photons emitted by the source in unit time increases
(b) the total energy of the photons emitted per unit time increases
(c) more energetic photons are emitted
(d) faster photons are emitted
An atom absorbs a photon of wavelength 500 nm and emits another photon of wavelength 700 nm. Find the net energy absorbed by the atom in the process.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The work function of a metal is 2.5 × 10−19 J. (a) Find the threshold frequency for photoelectric emission. (b) If the metal is exposed to a light beam of frequency 6.0 × 1014 Hz, what will be the stopping potential?
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
When a metal plate is exposed to a monochromatic beam of light of wavelength 400 nm, a negative potential of 1.1 V is needed to stop the photo current. Find the threshold wavelength for the metal.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Read the following paragraph and answer the questions.
| The figure shows the variation of photoelectric current measured in a photocell circuit as a function of the potential difference between the plates of the photocell when light beams A, B, C and D of different wavelengths are incident on the photocell. Examine the given figure and answer the following questions: |
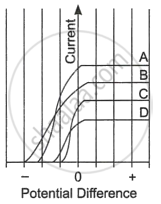
- Which light beam has the highest frequency and why?
- Which light beam has the longest wavelength and why?
- Which light beam ejects photoelectrons with maximum momentum and why?
How would the stopping potential for a given photosensitive surface change if the intensity of incident radiation was decreased? Justify your answer.
