Advertisements
Advertisements
प्रश्न
Consider a thin target (10–2 cm square, 10–3 m thickness) of sodium, which produces a photocurrent of 100 µA when a light of intensity 100W/m2 (λ = 660 nm) falls on it. Find the probability that a photoelectron is produced when a photons strikes a sodium atom. [Take density of Na = 0.97 kg/m3].
उत्तर
According to the problem, the area of the target A = 10–2 cm2 = 10–4 m2
And thickness, d = 10–3 m
Photocurrent, i = 100 × 10–6 A= 10–4 A
Intensity, I = 100 W/m2
⇒ λ = 660 nm = 660 × 10–9 m
ρNa = 0.97 kg/m3
Avogadro number = 6 × 1026 kg atom
Volume of sodium target = A × d
= 10–4 × 10–3
= 10–7 m3
We know that 6 × 1026 atoms of sodium weigh = 23 kg
Density of sodium = 0.97 kg/m3
Hence the volume of 6 × 1026 sodium atoms = `23/0.97` m3
Volume occupied by one sodium atom = `23/(0.97 xx (6 xx 10^36))` = 3.95 × 10–26 m3
Number of sodium atoms in target `(N_"sodium") = 10^-7/(3.95 xx 10^-26)` = 2.53 × 1018
Let m be the number of photons falling per second on the target.
Energy of each photon = `(hc)/A`
Total energy falling per second on target = `(nhc)/λ = IA`
∴ `n = (IAλ)/(hc)`
= `(100 xx 10^-4 xx (660 xx 10^-9))/((6.62 xx 10^-34) xx (3 xx 10^8))` = 3.3 × 1016
Let P be the probability of emission per atom per photon. The number of photoelectrons emitted per second
`N = P xx n xx (N_"sodium")`
= `P xx (33 xx 10^16) xx (2.53 xx 10^18)`
Now, according to the question,
i = 100 µA = 100 × 10–6 = 10–4 A
Current, i = Ne
∴ `10^-4 xx P xx (3.3 xx 10^16) xx (2.53 xx 10^18) xx (1.6 xx 10^-19)`
⇒ `P = 10^-4/((3.3 xx 10^16) xx (2.53 xx 10^18) xx (1.6 xx 10^-19))`
= 7.48 × 10–21
Then, the probability of photoemission by a single photon on a single atom is very much less than 1. Because the absorption of two photons by an atom is negligible.
APPEARS IN
संबंधित प्रश्न
It is found that photosynthesis starts in certain plants when exposed to sunlight, but it does not start if the plants are exposed only to infrared light. Explain.
Planck's constant has the same dimensions as
Two photons of
A point source causes photoelectric effect from a small metal plate. Which of the following curves may represent the saturation photocurrent as a function of the distance between the source and the metal?

A sphere of radius 1.00 cm is placed in the path of a parallel beam of light of large aperture. The intensity of the light is 0.5 W cm−2. If the sphere completely absorbs the radiation falling on it, find the force exerted by the light beam on the sphere.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The electric field associated with a monochromatic beam is 1.2 × 1015 times per second. Find the maximum kinetic energy of the photoelectrons when this light falls on a metal surface whose work function is 2.0 eV.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The figure is the plot of stopping potential versus the frequency of the light used in an experiment on photoelectric effect. Find (a) the ratio h/e and (b) the work function.
In photoelectric effect, the photoelectric current started to flow. This means that the frequency of incident radiations is ______.
Do all the electrons that absorb a photon come out as photoelectrons?
Read the following paragraph and answer the questions.
| The figure shows the variation of photoelectric current measured in a photocell circuit as a function of the potential difference between the plates of the photocell when light beams A, B, C and D of different wavelengths are incident on the photocell. Examine the given figure and answer the following questions: |
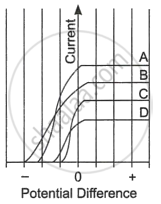
- Which light beam has the highest frequency and why?
- Which light beam has the longest wavelength and why?
- Which light beam ejects photoelectrons with maximum momentum and why?
