Advertisements
Advertisements
प्रश्न
Define the terms (i) ‘cut-off voltage’ and (ii) ‘threshold frequency’ in relation to the phenomenon of photoelectric effect.
Using Einstein’s photoelectric equation shows how the cut-off voltage and threshold frequency for a given photosensitive material can be determined with the help of a suitable plot/graph.
उत्तर
When light of sufficiently small wavelength is incident on a metal surface, electrons are ejected from the metal. This Phenomenon is called the photoelectric effect.
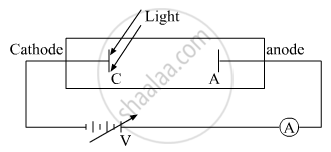
(i) The cathode is illuminated with light of some fixed frequency v and fixed Intensity I1. A small photoelectric current is observed due to few electrons that reach anode just because they have sufficiently large velocity of emission. If we made the potential of the anode negative with respect to cathode then the electrons emitted by cathode are repelled. Some electrons even go back to the cathode so that the current decreases. At a certain value of this negative potential, the current is completely stopped. The least value of this anode potential which just stops the photocurrent is called cut off potential or stopping potential.
(ii) For a given material, there is a certain minimum frequency that if the incident radiation has a frequency below this threshold, no photoelectric emission will take place, howsoever intense the radiation may be falling.
(iii) According to Einstein’s photoelectric equation, maximum K. E is given as
`K.E_(max) =(hc)/lambda phi = hv -phi`
Where λ is wavelength and ν is corresponding frequency and Φ is work function.
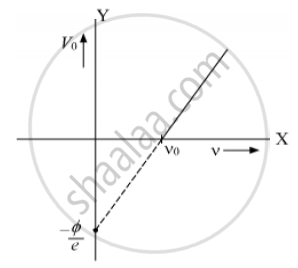
We expose a material to lights of various frequencies and thus photoelectric current is observed and cut off potential needed to reduce this current to Zero is noted. A graph is plotted and that is straight line.
At ν0 the stopping potential is zero this means at this frequency the incident light is not able to eject electrons this is threshold frequency. Extended this line gives the cut off potential to make the photo
current zero which is `(-phi)/e`
संबंधित प्रश्न
In an experiment on the photoelectric effect, the slope of the cut-off voltage versus the frequency of incident light is found to be 4.12 × 10−15 Vs. Calculate the value of Planck’s constant.
Briefly explain the three observed features which can be explained by Einstein’s photoelectric equation.
A non-monochromatic light is used in an experiment on photoelectric effect. The stopping potential
The electric field at a point associated with a light wave is `E = (100 "Vm"^-1) sin [(3.0 xx 10^15 "s"^-1)t] sin [(6.0 xx 10^15 "s"^-1)t]`.If this light falls on a metal surface with a work function of 2.0 eV, what will be the maximum kinetic energy of the photoelectrons?
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A monochromatic light source of intensity 5 mW emits 8 × 1015 photons per second. This light ejects photoelectrons from a metal surface. The stopping potential for this setup is 2.0 V. Calculate the work function of the metal.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A small metal plate (work function φ) is kept at a distance d from a singly-ionised, fixed ion. A monochromatic light beam is incident on the metal plate and photoelectrons are emitted. Find the maximum wavelength of the light beam, so that some of the photoelectrons may go round the ion along a circle.
In a photoelectric experiment, the collector plate is at 2.0 V with respect to the emitter plate made of copper (φ = 4.5 eV). The emitter is illuminated by a source of monochromatic light of wavelength 200 nm. Find the minimum and maximum kinetic energy of the photoelectrons reaching the collector.
Each photon has the same speed but different ______.
The wavelength of a photon needed to remove a proton from a nucleus which is bound to the nucleus with 1 MeV energy is nearly ______.
- In the explanation of photo electric effect, we assume one photon of frequency ν collides with an electron and transfers its energy. This leads to the equation for the maximum energy Emax of the emitted electron as Emax = hν – φ0 where φ0 is the work function of the metal. If an electron absorbs 2 photons (each of frequency ν) what will be the maximum energy for the emitted electron?
- Why is this fact (two photon absorption) not taken into consideration in our discussion of the stopping potential?
