Advertisements
Advertisements
प्रश्न
Describe one reaction of a carboxylic acid.
उत्तर
Ethanoic acid is a carboxylic acid which when reacts with ethanol (alcohol) produces sweet smelling ester. The process in which the carboxylic acid reacts with alcohol, in the presence of a catalyst, to form an ester is called esterification.
The chemical equation for the above reaction is as follows:
`CH_3 COOH +_2 H_5 OH` 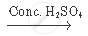 `CH_3 COOC_2 H_5 + H_2O`
`CH_3 COOC_2 H_5 + H_2O`
APPEARS IN
संबंधित प्रश्न
In an experiment to study the properties of ethanoic acid, a student takes about 3 mL of ethanoic acid in a dry test tube. He adds an equal amount of distilled water to it and shakes the test tube well. After some time he is likely to observe that
(A) a colloid is formed in the test tube.
(B) the ethanoic acid dissolves readily in water.
(C) the solution becomes light orange.
(D) water floats over the surface of ethanoic acid.
For preparing soap in the laboratory we require an oil and a base. Which of the following combinations of an oil and a base would be best suited for the preparation of soap?
(a) Castor oil and calcium hydroxide
(b) Turpentine oil and sodium hydroxide
(c) Castor oil and sodium hydroxide
(d) Mustard oil and calcium hydroxide
Give balanced chemical equations for Sodium ethanoate to methane.
When zinc powder is added to acetic acid ______________
(a) the mixture becomes warm
(b) a gas is evolved
(c) the colour of the mixture becomes yellow
(d) a solid settles at the bottom
Name the functional group present in an organic compound which gives brisk effervescence with NaHCO3.
Acetic acid is a typical acid. Write one equation in case of its reactions with a carbonate.
A student is studying the properties of acetic acid. List two physical properties of acetic acid he observes. What happens when he adds a pinch of sodium hydrogen carbonate to this acid? Write any two observations.
On adding acetic acid to sodium hydrogen carbonate in a test tube, a student observes
(A) no reaction
(B) a colourless gas with pungent smell
(C) bubbles of a colourless and odourless gas
(D) a strong smell of vinegar
Choose the correct option from given alternative:
When sodium hydrogen carbonate is added to ethanoic acid a gas evolves. Consider the following statements about the gas evolved?
(A) It turns lime water milky.
(B) It is evolved with brisk effervescence.
(C) It has a smell of burning sulfur.
(D) It is also a by-product of respiration.
Write the characteristics of ethanoic acid.
