Advertisements
Advertisements
Question
Describe one reaction of a carboxylic acid.
Solution
Ethanoic acid is a carboxylic acid which when reacts with ethanol (alcohol) produces sweet smelling ester. The process in which the carboxylic acid reacts with alcohol, in the presence of a catalyst, to form an ester is called esterification.
The chemical equation for the above reaction is as follows:
`CH_3 COOH +_2 H_5 OH` 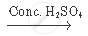 `CH_3 COOC_2 H_5 + H_2O`
`CH_3 COOC_2 H_5 + H_2O`
APPEARS IN
RELATED QUESTIONS
When you add about 2 ml of acetic acid to a test tube containing an equal amount of distilled water and leave the test tube to settle after shaking its contents, what will you observe in the test tube after about 5 minutes?
(A) A white precipitate settling at its bottom
(B) A clear colourless solution
(C) A layer of water over the layer of acetic acid
(D) A layer of acetic acid over the layer of water
In order to study saponification reaction, we first prepare 20% solution of sodium hydroxide. If we record the temperature of this solution just after adding sodium hydroxide flakes to water and also test its nature using litmus, it may be concluded that the process of making this solution is
(A) exothermic and the solution is alkaline
(B) endothermic and the solution is alkaline
(C) endothermic and the solution is acidic
(D) exothermic and the solution is acidic
Name the following:
The distinctive reaction that takes place when ethanol is treated with acetic acid.
Name the functional group present in an organic compound which gives brisk effervescence with NaHCO3.
What will you observe when dilute ethanoic acid and dilute hydrochloric acid are put on universal indicator paper, one by one? What does it show?
What is the physical state of CH3COOH?
Name two oxidising agents which can oxidise ethanol to ethanoic acid.
An organic compound X of molecular formula C2H4O2 gives brisk effervescence with sodium hydrogen carbonate. Give the name and formula of X.
Give any two uses of ethanoic acid.
The structural formula of an ester is :

Write the formula of the acid and the alcohol from which it is formed.
