Advertisements
Advertisements
प्रश्न
Write names and formulae of hydrocarbons containing a single and a double bond (one example for each). Give one characteristic chemical property of each.
उत्तर
Methane is a hydrocarbon which has a single bond. Its formula is CH4.
The characteristic chemical property of methane is the substitution reaction. It is a reaction in which one or more hydrogen atoms are replaced by some other atoms.
Chemical equation:
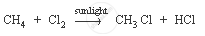
Ethene is a hydrocarbon containing a double bond. Its formula is C2H4.
The characteristic chemical property of ethene is the addition reaction. It is a reaction in which unsaturated compound (ethene) is combined with another substance to form a single product (saturated compound).
Chemical equation:
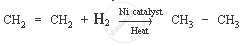
APPEARS IN
संबंधित प्रश्न
Fill in the following blank with suitable word.
The functional group present in ethanol is ...............
Write the name of the following functional group:

Consider the following organic compound:
CH3OH, C2H5OH, CH3COCH3, CH3COOH, C2H5COOH, C4H9COOC2H5, CH4, C2H6, CH3CHO, HCHO
Out of these compound:
Which compound is used to sterilise wounds and syringes?
How will you carry out the following conversions:
(i) Ethanol to ethene
(ii) Bromoethane to ethanol
[Ethane, Ethene, Ethanoic acid, Ethyne, Ethanol]
From the above list, name the compound with \[\ce{-OH}\] as the part of its structure.
What are the functional groups? Name any two compounds containing functional groups.
Write the molecular formula of the given compound.
Sodium ethoxide
What is the role of concentrated Sulphuric acid when it is heated with Ethanol at 443 K. Give the reaction involved.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
Give the balanced chemical equation of the following reaction:
Combustion of ethanol.
