Advertisements
Advertisements
प्रश्न
Write names and formulae of hydrocarbons containing a single and a double bond (one example for each). Give one characteristic chemical property of each.
उत्तर
Methane is a hydrocarbon which has a single bond. Its formula is CH4.
The characteristic chemical property of methane is the substitution reaction. It is a reaction in which one or more hydrogen atoms are replaced by some other atoms.
Chemical equation:
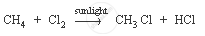
Ethene is a hydrocarbon containing a double bond. Its formula is C2H4.
The characteristic chemical property of ethene is the addition reaction. It is a reaction in which unsaturated compound (ethene) is combined with another substance to form a single product (saturated compound).
Chemical equation:
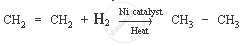
APPEARS IN
संबंधित प्रश्न
Fill in the blanks from the choices given within brackets:
The compound formed when ethanol reacts with sodium is____ (sodium ethanoate, sodium ethoxide, sodium propanoate).
Give a balanced equation for the preparation of ethanol from monochloroethane and aq. sodium hydroxide.
The catalyst used in the contact process is
(A) Copper
(B) Iron
(C) Vanadium pentoxide
(D) Manganese dioxide
Draw the structures for the following compounds of Pentanal
When ethanol reacts with sodium metal, it forms two products. These products are:
(a) sodium ethanaoate and oxygen
(b) sodium ethanaoate and hydrogen
(c) sodium ethoxide and oxygen
(d) sodium ethoxide and hydrogen
Write the equations of chlorination of ethane
At room temperature ethanol is _______.
How would you bring about the following conversions? Name the process and write the reaction involved.
Draw electron dot structure of Ethanol.
Give the balanced chemical equation of the following reaction:
Oxidation of ethanol by acidified potassium dichromate.
