Advertisements
Advertisements
प्रश्न
Differentiate between alkanes and alkenes. Name and draw the structure of one member of each.
उत्तर
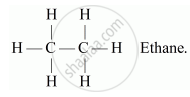
(a) Alkanes: These are the simplest hydrocarbons having general formula CnH2n+2, where n is the number of atoms. These compounds contain all single covalent bonds between the carbon atoms. Suffix-ane is used while naming alkanes.
For e.g. C2H6
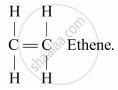
Alkenes: These are the simplest unsaturated hydrocarbons having general formula CnH2n, where n is the number of atoms. These compounds contain at least one double bond between the carbon atoms. Suffix, -ene is used while naming alkenes.
For e.g. C2H4
APPEARS IN
संबंधित प्रश्न
Give a chemical test to distinguish between saturated and unsaturated hydrocarbons.
How many structural isomers can you draw for pentane?
Draw the electron dot structures for H2S.
Write a short note on Catenation.
Giving their structures, state the number of single bonds, double bonds and triple bonds (if any) in the following compounds:
ethyne
An unsaturated hydrocarbon having a triple covalent bond has 50 hydrogen atoms in its molecule. The number of carbon atoms in its molecule will be:
(a) 24
(b) 25
(c) 26
(d) 28
Write the molecular formula of an alkene having 20 carbon atoms?
Which of the following hydrocarbons can decolourise bromine water and which cannot? Why?
C6H12, C6H14, C6H10
Answer the following question.
Name a cyclic unsaturated carbon compound.
Name the functional groups present in the following compounds
- CH3 CO CH2 CH2 CH2 CH3
- CH3 CH2 CH2 COOH
- CH3 CH2 CH2 CH2 CHO
- CH3 CH2 OH
