Advertisements
Advertisements
प्रश्न
Write a short note on Catenation.
उत्तर
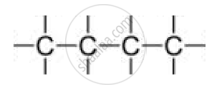
Catenation: The property of carbon element due to which its atoms can join one another to form long carbon chains is called catenation.
A) Straight chain of carbon atoms
B) Branched-chain of carbon atoms

C) Closed chain or ring chain of carbon atoms

APPEARS IN
संबंधित प्रश्न
Define Saturated hydrocarbon.
How many structural isomers can you draw for pentane?
What will be the formula and electron dot structure of cyclopentane?
Draw the electron dot structures for ethanoic acid.
Write the name and structure of a saturated compound in which the carbon atoms are arranged in a ring. Give the number of single bonds present in this compound.
Giving their structures, state the number of single bonds, double bonds and triple bonds (if any) in the following compounds:
ethyne
How many isomers of the following hydrocharbons are possible?
C3H8
Write the molecular formula of an alkene having 20 carbon atoms?
Which of the following hydrocarbons is unsaturated?
C3H4; C2H6
Name the reaction which is usually used in the conversion of vegetables oils to fats. Explain the reaction involved in detail. Write a chemical equation to illustrate your answer.
Fill in the blanks and rewrite the completed statements:
The organic compounds having double or triple bond in them are termed as _________________ _________________.
Explain the following reaction with balanced chemical equation:
Calcium reacts with water.
In which of the following compounds, — OH is the functional group?
Pentane has the molecular formula C5 H12 It has
Carbon, Group (14) element in the Periodic Table, is known to form compounds with many elements. Write an example of a compound formed with
- chlorine (Group 17 of Periodic Table)
- oxygen (Group 16 of Periodic Table)
Unsaturated hydrocarbons contain multiple bonds between the two C-atoms and show addition reactions. Give the test to distinguish ethane from ethene.
Name the reaction which is commonly used in the conversion of vegetable oils to fats. Explain the reaction involved in detail.
Write the name of the following compound
\[\begin{array}{cc}
\phantom{}\ce{H}\phantom{...}\ce{H}\phantom{...}\ce{H}\phantom{.....}\\
\phantom{}|\phantom{....}|\phantom{....}|\phantom{.....}\\
\ce{H} - \ce{C} - \ce{C} - \ce{C} ≡ \ce{C} - \ce{H}\\
\phantom{}|\phantom{....}|\phantom{..........}\\
\phantom{}\ce{H}\phantom{...}\ce{H}\phantom{..........}\\
\end{array}\]
The formulae of four organic compounds are shown below. Choose the correct option
| A | B | C | D |
| \[\begin{array}{cc} \phantom{.}\ce{H}\phantom{......}\ce{H}\\ \phantom{.}\backslash\phantom{.....}/\\ \ce{C = C}\\ /\phantom{.....}\backslash\\ \ce{H}\phantom{......}\ce{H} \end{array}\] |
\[\begin{array}{cc} \phantom{........}\ce{H}\phantom{.....}\ce{O}\\ \phantom{.......}|\phantom{....}//\\ \ce{H - C - C}\\ \phantom{......}|\phantom{.....}\backslash\\ \phantom{...........}\ce{H}\phantom{.....}\ce{O - H} \end{array}\] |
\[\begin{array}{cc}
|
\[\begin{array}{cc} \ce{H}\phantom{...}\ce{H}\phantom{....}\\ |\phantom{....}|\phantom{....}\\ \ce{H - C - C - O - H}\\ |\phantom{....}|\phantom{.....}\\ \ce{H}\phantom{....}\ce{H}\phantom{.....} \end{array}\] |
